15 May, 2020 COVID-19 continues to disrupt clinical trials, with 69.9% of disruptions due to the suspension of enrolment
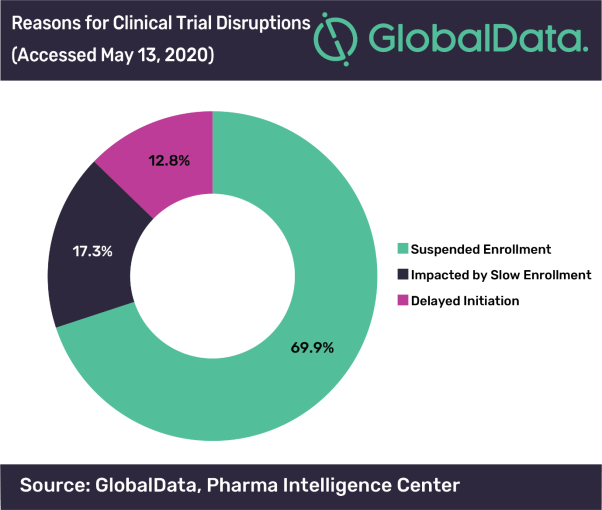
Posted in PharmaClinical trials across the globe continue to be disrupted due to COVID-19. According to GlobalData, a leading data and analytics company, among global clinical trials that have been disrupted since April 2020, 69.9% are due to the suspension of enrolment, 17.3% due to slow enrolment and 12.8% due to delayed initiation. Furthermore, within the 17.3% of trials affected by slow enrolment, 15.4% are specifically due to the availability of sites and investigators.
Brooke Wilson, Associate Director of Trials Intelligence at GlobalData, comments: “Many hospitals that serve as trial sites are being inundated with COVID-19 patients and are no longer available. For that same reason, many investigators may be repurposed to COVID-19 drug discovery trials or treating COVID-19 patients, or activation of sites for non-COVID-19 trials are being deprioritized. There is also a high risk to subjects in a clinical trial that have a serious chronic or acute condition that affects their immune system, giving them a greater chance of contracting COVID-19.
“There are also now over 60 trials that were previously disrupted from the COVID-19 pandemic by delayed initiation and slow or suspended enrolment that are now ongoing or completed. The upward trend for clinical trial delays may begin to slow moving forward as more companies adjust, along with a possible shift toward virtual trials.”
While clinical trials research is being delayed due to COVID-19, the FDA has issued some industry guidance to help trials proceed during the pandemic.
Brooke added: “The methods outlined by the FDA to help trials proceed during the pandemic, including using alternative contact methods such as virtual visits and phone calls, as well as the use of self-administration and remote monitoring, will help with the issues of quarantine, travel limitations, clinical site closures and disrupted supply chains.”
