Community Acquired Pneumonia Drugs in Development by Stages, Target, MoA, RoA, Molecule Type and Key Players, 2022 Update
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
Community-acquired pneumonia (CAP) is defined as pneumonia acquired outside a hospital or long-term care facility. Symptoms include cough, fever, shaking chills, confusion, headache and loss of appetite. Risk factors include chronic lung disease, cigarette smoking, dementia, stroke, brain injury, heart disease, liver cirrhosis and diabetes mellitus. Treatment includes antibiotics and healthy lifestyle.
The Community-acquired Pneumonia Drugs in Development market research report provides an in-depth analysis of the Community-acquired Pneumonia pipeline landscape. The report provides comprehensive information on the therapeutics under development for Community-Acquired Pneumonia, complete with analysis by stage of development, drug target, mechanism of action (MoA), route of administration (RoA), and molecule type. The report also covers the descriptive pharmacological action of the therapeutics, its complete research and development history, and the latest news and press releases. Additionally, the report provides an overview of key players involved in therapeutic development for Community Acquired Pneumonia and features dormant and discontinued projects.
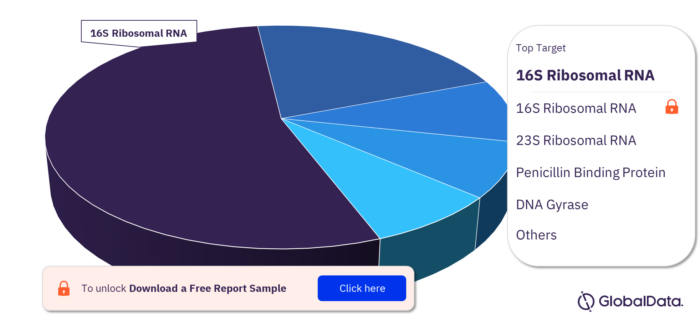
Community-acquired Pneumonia Pipeline Drugs Market Targets
The targets of the Community-acquired Pneumonia pipeline drugs market are 16S Ribosomal RNA, 23S Ribosomal RNA, Penicillin Binding Protein, DNA Gyrase, DNA Topoisomerase IV, 30S Ribosomal Subunit, Complement C5, Gelsolin, Interferon Alpha/Beta Receptor 1, Interferon Alpha/Beta Receptor 2, and Penicillin Binding Protein 2a.
Community-acquired Pneumonia Pipeline Drugs Market, By Targets
For more target insights, download a free report sample
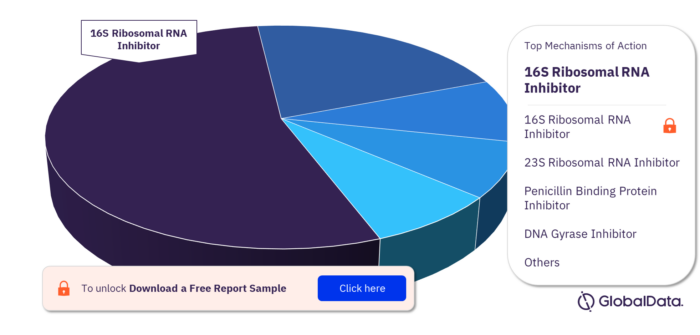
Mechanisms of Action of Community-Acquired Pneumonia Pipeline Drugs Market
The mechanisms of action of the Community-acquired Pneumonia pipeline drugs market are 16S Ribosomal RNA Inhibitor, 23S Ribosomal RNA Inhibitor, Penicillin Binding Protein Inhibitor, DNA Gyrase Inhibitor, DNA Topoisomerase IV Inhibitor, 30S Ribosomal Subunit Inhibitor, Complement C5 Inhibitor, Gelsolin Replacement, Interferon Alpha/Beta Receptor 1 Agonist, Interferon Alpha/Beta Receptor 2 Agonist, and Penicillin Binding Protein 2a Inhibitor.
Community-acquired Pneumonia Pipeline Drugs Market, By Mechanisms of Action
For more mechanisms of action insights, download a free report sample
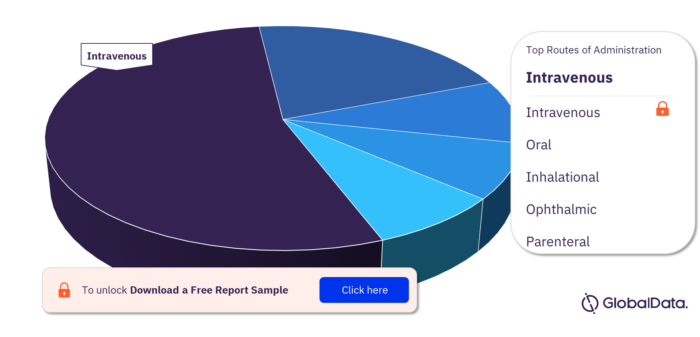
Routes of Administration in Community-Acquired Pneumonia Pipeline Drugs Market
The routes of administration in the Community-acquired Pneumonia pipeline drugs market are intravenous, oral, inhalational, ophthalmic, and parenteral.
Community-acquired Pneumonia Pipeline Drugs Market, By Routes of Administration
For more routes of administration insights, download a free report sample
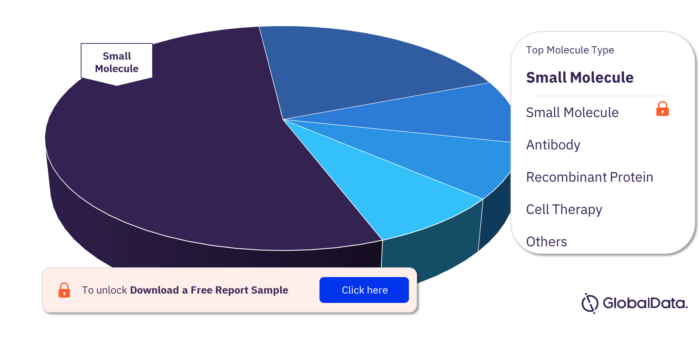
Molecule Types in Community-Acquired Pneumonia Pipeline Drugs Market
The molecule types in the Community-acquired Pneumonia pipeline drugs market are small molecule, antibody, recombinant protein, cell therapy, monoclonal antibody, and protein.
Community-acquired Pneumonia Pipeline Drugs Market, By Molecule Types
For more molecule type insights, download a free report sample
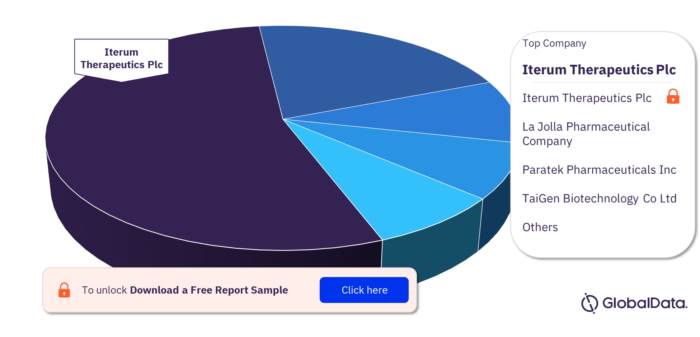
Key Community-Acquired Pneumonia Pipeline Drugs Market Companies
Some of the key companies in the Community-acquired Pneumonia pipeline drugs market are Iterum Therapeutics Plc, La Jolla Pharmaceutical Company, Paratek Pharmaceuticals Inc, TaiGen Biotechnology Co Ltd, Takeda Pharmaceutical Co Ltd, Wockhardt Ltd, Beijing FuKangren Bio-pharm Tech Co Ltd, BioAegis Therapeutics Inc, Biotest AG, and Eagle Pharmaceuticals Inc.
Community-acquired Pneumonia Pipeline Drugs Market, By Key Companies
To know more about key companies, download a free report sample
Market report overview
| Targets | 16S Ribosomal RNA, 23S Ribosomal RNA, Penicillin Binding Protein, DNA Gyrase, DNA Topoisomerase IV, 30S Ribosomal Subunit, Complement C5, Gelsolin, Interferon Alpha/Beta Receptor 1, Interferon Alpha/Beta Receptor 2, and Penicillin Binding Protein 2a |
| Mechanisms of Action | 16S Ribosomal RNA Inhibitor, 23S Ribosomal RNA Inhibitor, Penicillin Binding Protein Inhibitor, DNA Gyrase Inhibitor, DNA Topoisomerase IV Inhibitor, 30S Ribosomal Subunit Inhibitor, Complement C5 Inhibitor, Gelsolin Replacement, Interferon Alpha/Beta Receptor 1 Agonist, Interferon Alpha/Beta Receptor 2 Agonist, and Penicillin Binding Protein 2a Inhibitor |
| Routes of Administration | Intravenous, Oral, Inhalational, Ophthalmic, and Parenteral |
| Molecule Types | Small Molecule, Antibody, Recombinant Protein, Cell Therapy, Monoclonal Antibody, and Protein |
| Key Companies | Iterum Therapeutics Plc, La Jolla Pharmaceutical Company, Paratek Pharmaceuticals Inc, TaiGen Biotechnology Co Ltd, Takeda Pharmaceutical Co Ltd, Wockhardt Ltd, Beijing FuKangren Bio-pharm Tech Co Ltd, BioAegis Therapeutics Inc, Biotest AG, and Eagle Pharmaceuticals Inc |
Scope
This report provides:
- A snapshot of the global therapeutic landscape of Community-acquired Pneumonia (Infectious Disease).
- Reviews of pipeline therapeutics for Community-acquired Pneumonia (Infectious Disease) by companies and universities/research institutes based on information derived from company and industry-specific sources.
- Pipeline products based on various stages of development ranging from pre-registration to discovery and undisclosed stages.
- Descriptive drug profiles for the pipeline products which include product description, descriptive MoA, R&D brief, licensing and collaboration details & other developmental activities.
- Reviews of key companies involved in Community-acquired Pneumonia (Infectious Disease) therapeutics and enlists all their major and minor projects.
- Evaluation of Community-acquired Pneumonia (Infectious Disease) targeted therapeutics based on drug target, mechanism of action (MoA), route of administration (RoA), and molecule type.
- All the dormant and discontinued pipeline projects.
- Reviews of the latest news and deals related to pipeline therapeutics for Community-acquired Pneumonia (Infectious Disease).
Reasons to Buy
- Gain strategically significant competitor information, analysis, and insights to formulate effective R&D strategies.
- Identify emerging players with potentially strong product portfolio and create effective counter-strategies to gain competitive advantage.
- Identify and understand the targeted therapy areas and indications for Community-acquired Pneumonia (Infectious Disease). Identify the use of drugs for target identification and drug repurposing.
- Identify potential new clients or partners in the target demographic.
- Develop strategic initiatives by understanding the focus areas of leading companies.
- Plan mergers and acquisitions effectively by identifying key players and its most promising pipeline therapeutics.
- Devise corrective measures for pipeline projects by understanding Community-acquired Pneumonia (Infectious Disease) pipeline depth and focus of Indication therapeutics.
- Develop and design in-licensing and out-licensing strategies by identifying prospective partners with the most attractive projects to enhance and expand business potential and scope.
BioAegis Therapeutics Inc
Biotest AG
Eagle Pharmaceuticals Inc
Guangzhou Baiyunshan Pharmaceutical Holdings Co Ltd
InflaRx NV
ioGenetics Inc
Iterum Therapeutics Plc
Korea Pharma Co Ltd
La Jolla Pharmaceutical Company
Meiji Seika Pharma Co Ltd
Merck & Co Inc
Nabriva Therapeutics Plc
Paratek Pharmaceuticals Inc
ProThera Biologics Inc
TaiGen Biotechnology Co Ltd
Takeda Pharmaceutical Co Ltd
Wockhardt Ltd
Table of Contents
Table
Figures
Frequently asked questions
-
What are the targets of the Community-acquired Pneumonia pipeline drugs market?
The targets of the Community-acquired Pneumonia pipeline drugs market are 16S Ribosomal RNA, 23S Ribosomal RNA, Penicillin Binding Protein, DNA Gyrase, DNA Topoisomerase IV, 30S Ribosomal Subunit, Complement C5, Gelsolin, Interferon Alpha/Beta Receptor 1, Interferon Alpha/Beta Receptor 2, and Penicillin Binding Protein 2a.
-
What are the mechanisms of action of the Community-acquired Pneumonia pipeline drugs market?
The mechanisms of action of the Community-acquired Pneumonia pipeline drugs market are 16S Ribosomal RNA Inhibitor, 23S Ribosomal RNA Inhibitor, Penicillin Binding Protein Inhibitor, DNA Gyrase Inhibitor, DNA Topoisomerase IV Inhibitor, 30S Ribosomal Subunit Inhibitor, Complement C5 Inhibitor, Gelsolin Replacement, Interferon Alpha/Beta Receptor 1 Agonist, Interferon Alpha/Beta Receptor 2 Agonist, and Penicillin Binding Protein 2a Inhibitor.
-
What are the routes of administration in the Community-acquired Pneumonia pipeline drugs market?
The routes of administration in the Community-acquired Pneumonia pipeline drugs market are intravenous, oral, inhalational, ophthalmic, and parenteral.
-
What are the molecule types in the Community-acquired Pneumonia pipeline drugs market?
The molecule types in the Community-acquired Pneumonia pipeline drugs market are small molecule, antibody, recombinant protein, cell therapy, monoclonal antibody, and protein.
-
Which are the key companies in the Community-acquired Pneumonia pipeline drugs market?
Some of the key companies in the Community-acquired Pneumonia pipeline drugs market are Iterum Therapeutics Plc, La Jolla Pharmaceutical Company, Paratek Pharmaceuticals Inc, TaiGen Biotechnology Co Ltd, Takeda Pharmaceutical Co Ltd, Wockhardt Ltd, Beijing FuKangren Bio-pharm Tech Co Ltd, BioAegis Therapeutics Inc, Biotest AG, and Eagle Pharmaceuticals Inc.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more Infectious Disease reports