Influenza A Virus, H3N2 Subtype Infections Drugs in Development by Stages, Target, MoA, RoA, Molecule Type and Key Players, 2022 Update
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
H3N2 infections are caused by variant H3N2 virus which is an influenza virus. Symptoms include cough, sore throat, runny or stuffy nose, chills, nausea, vomiting, and/or diarrhea. Risk factors include age, chronic medical conditions like asthma, diabetes, heart disease, weakened immune systems, and neurological or neurodevelopment conditions. Treatment includes antiviral medications and antibiotics.
The Influenza A Virus, H3N2 – drugs in development research report provides a comprehensive overview on the therapeutics under development for Influenza A Virus, H3N2, complete with analysis by stage of development, drug target, mechanism of action (MoA), route of administration (RoA) and molecule type. The report also covers the descriptive pharmacological action of the therapeutics, its complete research and development history and latest news and press releases. Additionally, the report provides an overview of key players involved in therapeutic development for Influenza A Virus, H3N2 and features dormant and discontinued projects.
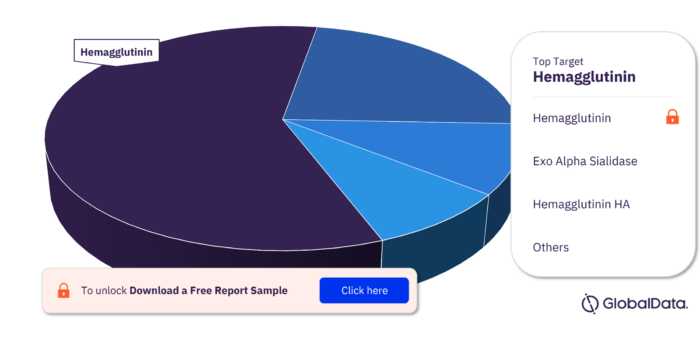
Key Targets in the Influenza A Virus, H3N2 Pipeline Drugs Market
The key targets in the Influenza A Virus, H3N2 pipeline drugs market are Hemagglutinin, Exo Alpha Sialidase, Hemagglutinin HA, 2019 Novel corona Virus Spike Glycoprotein, Hemagglutinin 1, Lipopolysaccharide, Substance P Receptor, Toll Like Receptor 2, Toll Like Receptor 3, and Toll Like Receptor 6.
Influenza A Virus, H3N2 Drugs Market, by Targets
For more target insights, download a free report sample
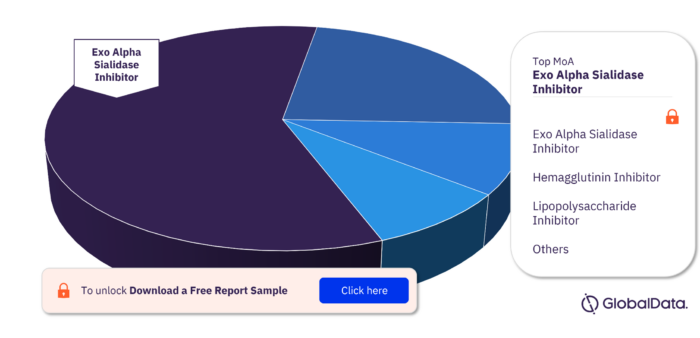
Key MoA in the Influenza A Virus, H3N2 Pipeline Drugs Market
The key mechanisms of action in the Influenza A Virus, H3N2 pipeline drugs market are Exo Alpha Sialidase Inhibitor, Hemagglutinin Inhibitor, Lipopolysaccharide Inhibitor, Substance P Receptor Agonist, Toll Like Receptor 2 Agonist, Toll Like Receptor 3 Agonist, and Toll Like Receptor 6 Agonist.
Influenza A Virus, H3N2 Pipeline Drugs Market, by MoA
To get more insights on key MoA, download a free sample report
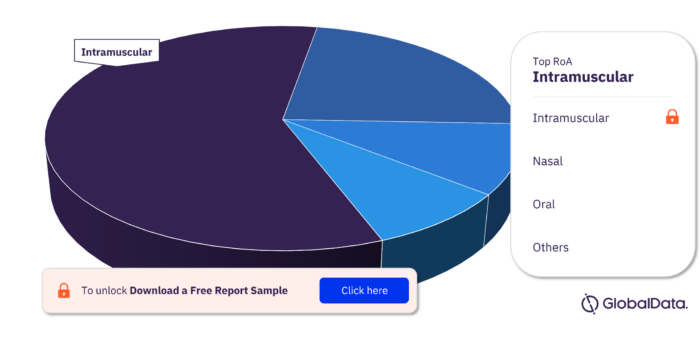
Influenza A Virus, H3N2 Pipeline Drugs Market Segmentation by RoA
The key routes of administration in the Influenza A Virus, H3N2 pipeline drugs market are intramuscular, nasal, oral, intravenous, subcutaneous, inhalational, intradermal, parenteral, and transdermal.
Influenza A Virus, H3N2 Pipeline Drugs Market Analysis, by RoA
To get more insights on key RoA, download a free sample report
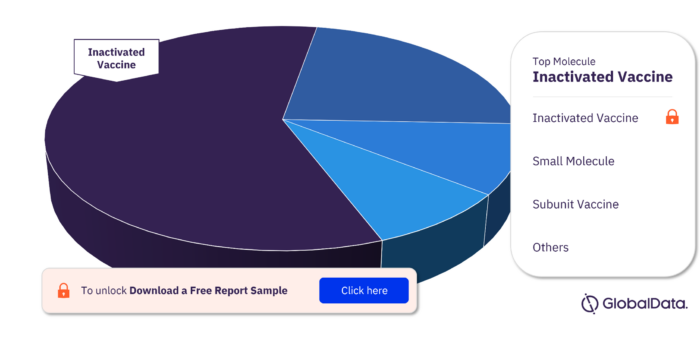
Key Molecule Types in the Influenza A Virus, H3N2 Pipeline Drugs Market
The key molecule types in the Influenza A Virus, H3N2 pipeline drugs market are Inactivated Vaccine, Small Molecule, Subunit Vaccine, Live Attenuated Vaccine, mRNA Vaccine, Recombinant Vector Vaccine, Monoclonal Antibody, Antibody, DNA Vaccine, and Fusion Protein among others.
Influenza A Virus, H3N2 Pipeline Drugs Market, by Molecule Type
To get more insights on key molecule types, download a free sample report
Major Companies in the Influenza A Virus, H3N2 Pipeline Drugs Market
The major companies in the Influenza A Virus, H3N2 pipeline drugs market are Sanofi, Adimmune Corp, FluGen Inc, Aphios Corp, CSL Ltd, Moderna Inc, NanoViricides Inc, and AbbVie Inc among others.
Influenza A Virus, H3N2 Pipeline Drugs Market, by Major Companies
For more company insights, download a free sample report
Influenza A Virus, H3N2 Pipeline Drugs Market Overview
| Key Targets | Exo Alpha Sialidase, Hemagglutinin HA, 2019 Novel corona Virus Spike Glycoprotein, Hemagglutinin 1, Lipopolysaccharide, Substance P Receptor, Toll Like Receptor 2, Toll Like Receptor 3, and Toll Like Receptor 6 |
| Key Mechanisms of Action | Exo Alpha Sialidase Inhibitor, Hemagglutinin Inhibitor, Lipopolysaccharide Inhibitor, Substance P Receptor Agonist, Toll Like Receptor 2 Agonist, Toll Like Receptor 3 Agonist, and Toll Like Receptor 6 Agonist |
| Key Routes of Administration | Intramuscular, Nasal, Oral, Intravenous, Subcutaneous, Inhalational, Intradermal, Parenteral, and Transdermal |
| Key Molecule Types | Monoclonal Antibody, Small Molecule, Recombinant Protein, Fusion Protein, Modified Cell Therapy, Synthetic Peptide, and Biologic |
| Major Companies | Sanofi, Adimmune Corp, FluGen Inc, Aphios Corp, CSL Ltd, Moderna Inc, NanoViricides Inc, and AbbVie Inc |
Scope
- The pipeline guide provides a snapshot of the global therapeutic landscape of Influenza A Virus, H3N2
- The pipeline guide reviews pipeline therapeutics for Influenza A Virus, H3N2 by companies and universities/research institutes based on information derived from company and industry-specific sources.
- The pipeline guide covers pipeline products based on several stages of development ranging from pre-registration till discovery and undisclosed stages.
- The pipeline guide features descriptive drug profiles for the pipeline products which comprise, product description, descriptive licensing and collaboration details, R&D brief, MoA & other developmental activities.
- The pipeline guide reviews key companies involved in Influenza A Virus, H3N2 therapeutics and enlists all their major and minor projects.
- The pipeline guide evaluates Influenza A Virus, H3N2 therapeutics based on mechanism of action (MoA), drug target, route of administration (RoA), and molecule type.
- The pipeline guide encapsulates all the dormant and discontinued pipeline projects.
- The pipeline guide reviews the latest news related to pipeline therapeutics for Influenza A Virus, H3N2
Reasons to Buy
- Procure strategically important competitor information, analysis, and insights to formulate effective R&D strategies.
- Recognize emerging players with a potentially strong product portfolio and create effective counter-strategies to gain a competitive advantage.
- Find and recognize significant and varied types of therapeutics under development for Influenza A Virus, H3N2
- Classify potential new clients or partners in the target demographic.
- Develop tactical initiatives by understanding the focus areas of leading companies.
- Plan mergers and acquisitions meritoriously by identifying key players and their most promising pipeline therapeutics.
- Formulate corrective measures for pipeline projects by understanding Influenza A Virus, H3N2 pipeline depth and focus of Indication therapeutics.
- Develop and design in-licensing and out-licensing strategies by identifying prospective partners with the most attractive projects to enhance and expand business potential and scope.
- Adjust the therapeutic portfolio by recognizing discontinued projects and understand from the know-how what drove them from the pipeline.
Adimmune Corp
Alla Chem LLC
Allergy Therapeutics Plc
AlphaVax Inc
Ansun Biopharma Inc
Aphios Corp
AstraZeneca Plc
AusBio Ltd
BiondVax Pharmaceuticals Ltd
BlueWillow Biologics Inc
Brandenburg Antiinfektiva GmbH
Celltrion Inc
Changchun Bcht Biotechnology Co Ltd
Chicago Biosolutions Inc
Cidara Therapeutics Inc
CSL Ltd
Emergent BioSolutions Inc
Ena Respiratory Pty Ltd
FluGen Inc
Gamma Vaccines Pty Ltd
ILiAD Biotechnologies LLC
Inovio Pharmaceuticals Inc
InvVax Inc
Jiangsu Ab&b Biotechnology Co Ltd
Jiangsu Kanion Pharmaceutical Co Ltd
Liaoning Cheng Da Biotechnology Co Ltd
Moderna Inc
NanoViricides Inc
New Amsterdam Sciences Inc
Novavax Inc
PrEP Biopharm Ltd
Sanofi
Seqirus Ltd
Serum Institute of India Pvt Ltd
Shanghai Institute of Biological Products Co Ltd
Trellis Bioscience Inc
Vacthera BioTech GmbH
Zosano Pharma Corp
Table of Contents
Table
Figures
Frequently asked questions
-
What are the key targets in the Influenza A Virus, H3N2 pipeline drugs market?
The key targets in the Influenza A Virus, H3N2 pipeline drugs market are Exo Alpha Sialidase, Hemagglutinin HA, 2019 Novel corona Virus Spike Glycoprotein, Hemagglutinin 1, Lipopolysaccharide, Substance P Receptor, Toll Like Receptor 2, Toll Like Receptor 3, and Toll Like Receptor 6.
-
What are the key mechanisms of action in the Influenza A Virus, H3N2 pipeline drugs market?
The key mechanisms of action in the Influenza A Virus, H3N2 pipeline drugs market are Exo Alpha Sialidase Inhibitor, Hemagglutinin Inhibitor, Lipopolysaccharide Inhibitor, Substance P Receptor Agonist, Toll Like Receptor 2 Agonist, Toll Like Receptor 3 Agonist, and Toll Like Receptor 6 Agonist.
-
What are the key routes of administration in the Influenza A Virus, H3N2 pipeline drugs market?
The key routes of administration in the Influenza A Virus, H3N2 pipeline drugs market are intramuscular, nasal, oral, intravenous, subcutaneous, inhalational, intradermal, parenteral, and transdermal.
-
What are the key molecule types in the Influenza A Virus, H3N2 pipeline drugs market?
The key molecule types in the Influenza A Virus, H3N2 pipeline drugs market are Inactivated Vaccine, Small Molecule, Subunit Vaccine, Live Attenuated Vaccine, mRNA Vaccine, Recombinant Vector Vaccine, Monoclonal Antibody, Antibody, DNA Vaccine, and Fusion Protein.
-
What are the major companies in the Influenza A Virus, H3N2 pipeline drugs market?
The major companies in the Influenza A Virus, H3N2 pipeline drugs market are Sanofi, Adimmune Corp, FluGen Inc, Aphios Corp, CSL Ltd, Moderna Inc, NanoViricides Inc, and AbbVie Inc.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more Infectious Disease reports