Clinical Trials – The Expanding Landscape of Regenerative Medicine
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
Regenerative Medicine Clinical Trials Market Overview
Regenerative medicine can be defined as the development and application of treatments that promote the process of replacing, engineering, or regenerating human or animal tissues to restore or establish normal function lost due to aging and disease. Regenerative medicine in GlobalData’s Pharma Intelligence Center is derived based on the FDA’s regenerative medicine advanced therapy (RMAT) designation or designated as such per regulatory authorities. As technology advancement continues to grow each year, so does the usage of regenerative medicine. This report provides an analysis of regenerative medicine clinical trial data and trends through the years.
The greatest number of regenerative medicine clinical trials occurred in 2020. This increase could be because of an improvement in funding for cell and gene therapy. North America and the US-dominated regenerative medicine clinical trials by geography. However, for industry-sponsored trials, Mesoblast topped regenerative medicine trials.
| Key Regions | North America, Asia-Pacific, Europe, South and Central America, and The Middle East And Africa |
| Key Therapy Areas | Cardiovascular, Central Nervous System, Oncology, Gastrointestinal, Infectious Disease, Immunology, Metabolic Disorders, and Ophthalmology |
| Key Termination Reasons | Low Accrual Rate, Business/Strategic Decisions, Financial, Product Discontinuation, and Lack of Efficacy |
| Leading Industry Sponsors | Mesoblast, Novartis, Anterogen, Pharmicell, Helixmith, and Anges |
| Leading Non-Industry Sponsors | The Foundation for Orthopaedics And Regenerative Medicine, University of Pennsylvania, Samsung Medical Center, University of Miami, Mayo Clinic, And Standford University |
| Key Drugs | Novartis’s Tisagenlecleucel, Fludarabine, ISN-001, Stempeucel, and Allorx Stem Cell Therapy |
| Enquire & Decide | Discover the perfect solution for your business needs. Enquire now and let us help you make an informed decision before making a purchase. |
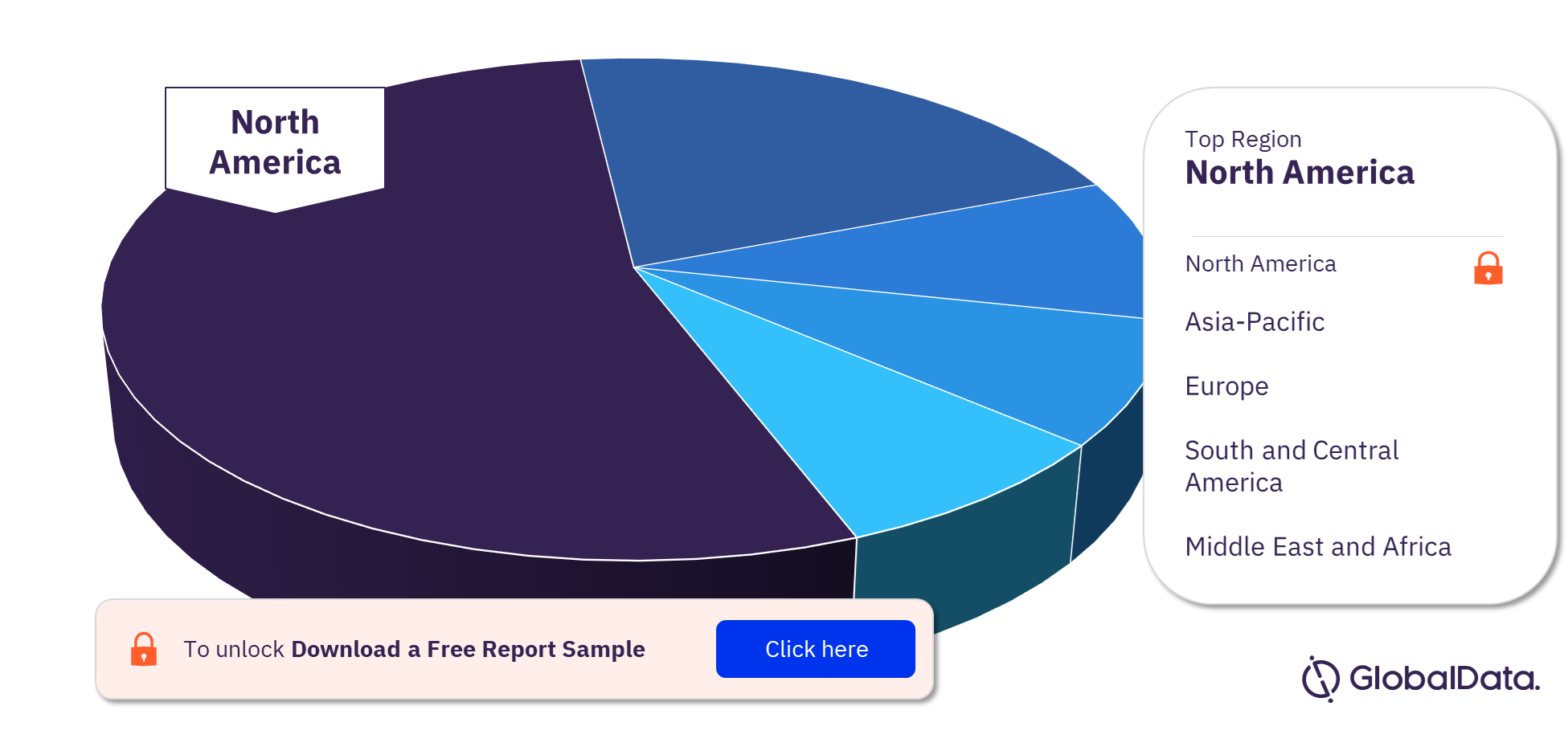
Regenerative Medicine Clinical Trials by Regions
Most regenerative clinical trials have been conducted in North America. This is followed by Asia-Pacific, Europe, South and Central America, and the Middle East and Africa. In Asia-Pacific, South Korea has conducted the highest number of regenerative medicine clinical trials.
Many countries such as Germany, Austria, and Ireland have a ban on regenerative medicine studies such as embryonic stem cell research, leading to a greater number of single-country trials. Regenerative medicine is a major challenge in developing countries. Due to financial limitations and ethical beliefs in the benefits of out-of-date therapies.
Regenerative Medicine Clinical Trials Analysis by Regions, 2022 (%)
To know more about the key regions for regenerative medicine clinical trials, download a free report sample.
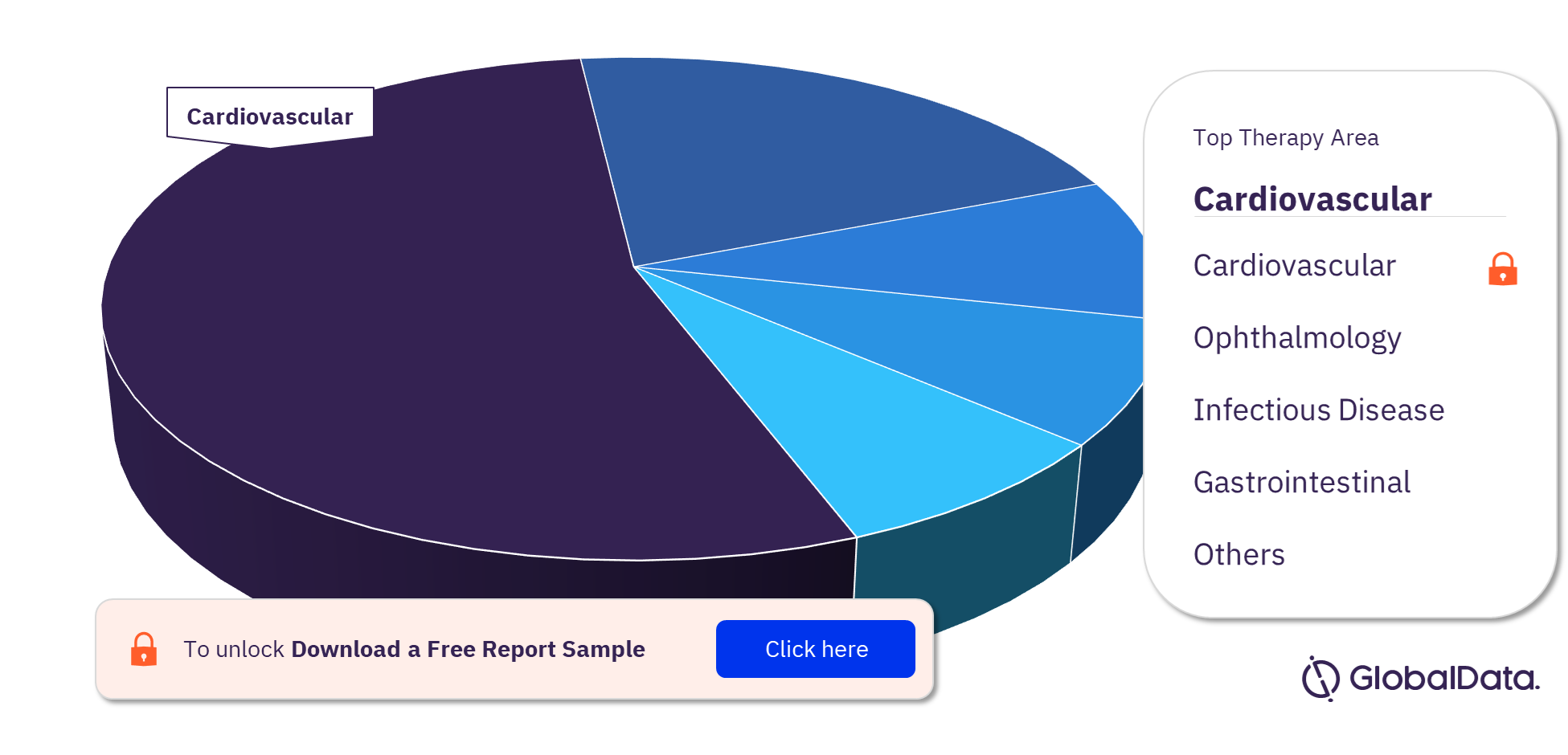
Regenerative Medicine Clinical Trials by Therapy Areas
Cardiovascular is the most researched therapy area when looking at regenerative medicine clinical trials. This is followed by central nervous system and oncology. Other key therapy areas of regenerative medicine clinical trials are gastrointestinal, infectious disease, immunology, metabolic disorders, and ophthalmology among others.
Regenerative Medicine Clinical Trials Analysis by Therapy Areas, 2022 (%)
To know more about the key therapy areas of regenerative medicine clinical trials, download a free report sample.
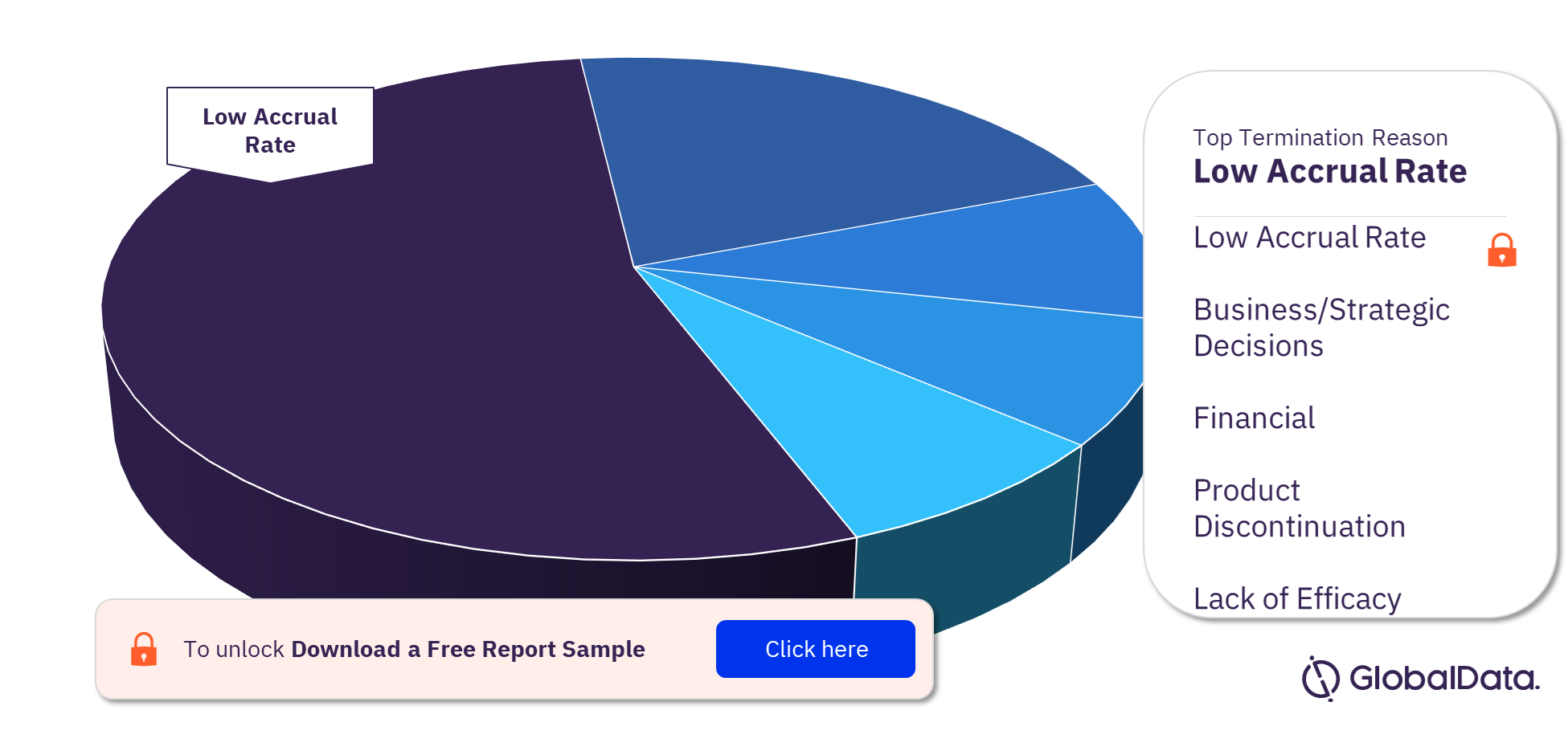
Regenerative Medicine Clinical Trials by Termination Reasons
Low accrual rate is the primary cause of termination for regenerative medicine clinical trials. This may be due to the notion of it having the highest risk of failure due to the increase in unapproved regenerative medicine. The other significant termination reasons are business/strategic decisions, financial, and product discontinuation. Lack of efficacy is number five for regenerative medicine.
Regenerative Medicine Clinical Trials Analysis, by Termination Reasons, 2022 (%)
To know more about the key termination reasons for regenerative medicine clinical trials, download a free report sample.
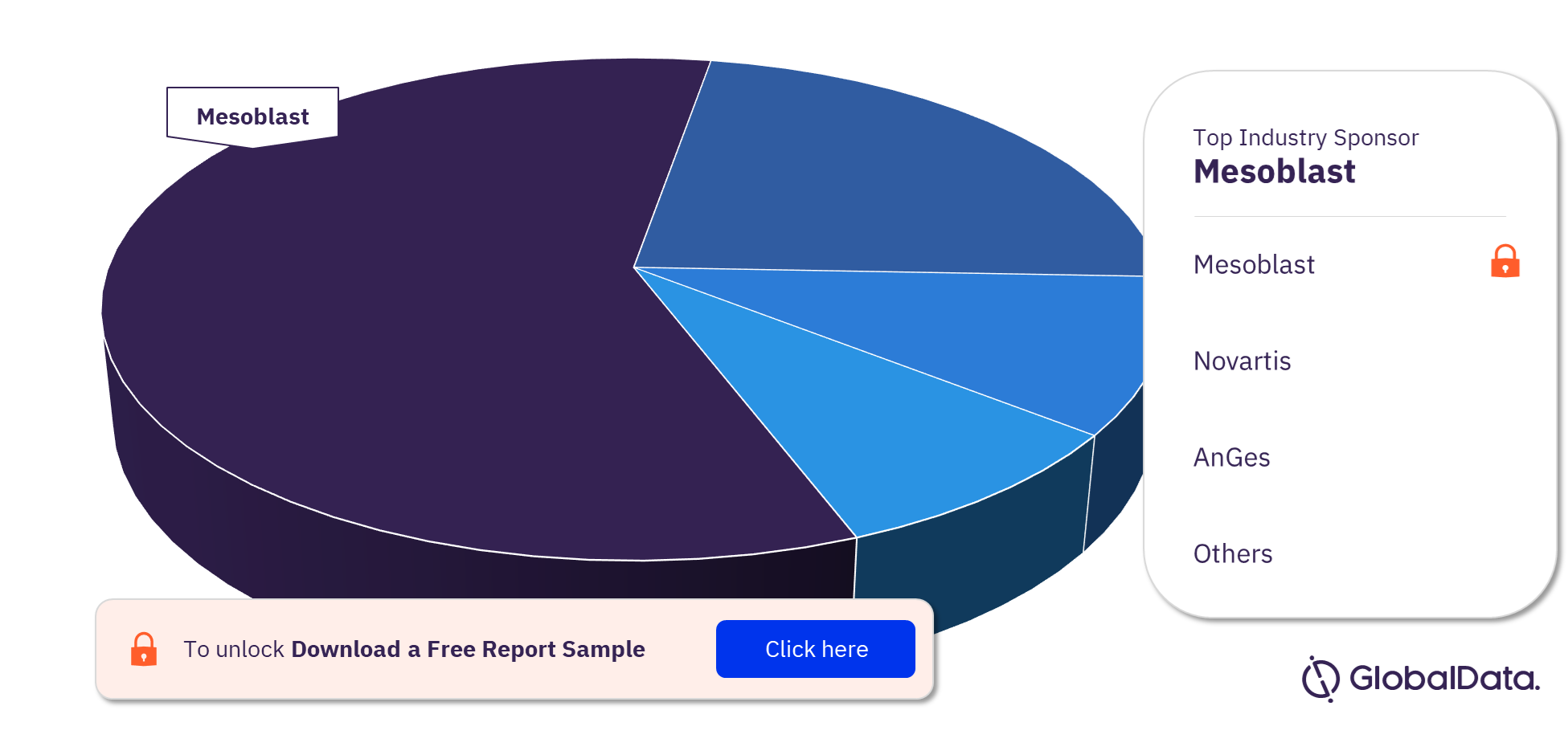
Regenerative Medicine Clinical Trials by Top Industry Sponsors
Mesoblast is the top industry sponsor with the most regenerative medicine clinical trials, followed by Novartis and Anterogen. Mesoblast has the most completed trials while Anterogen has the most ongoing and Novartis has the most planned trials. Other leading industry sponsors are Pharmicell, Helixmith, and AnGes among others.
Regenerative Medicine Clinical Trials Analysis, by Top Industry Sponsors, 2022 (%)
To know more about the leading industry sponsors of regenerative medicine clinical trials, download a free report sample.
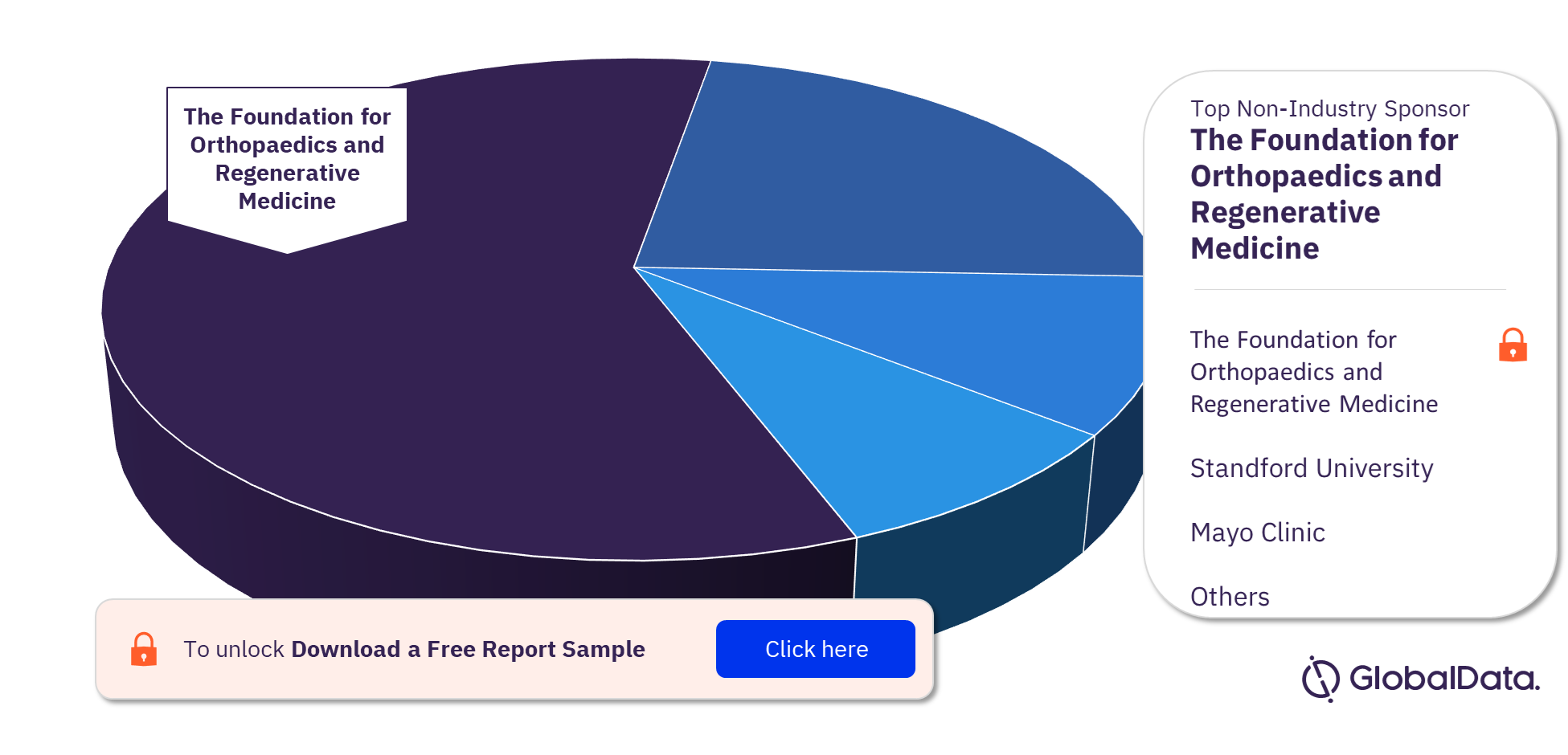
Regenerative Medicine Clinical Trials by Top Non-Industry Sponsors
The Foundation for Orthopaedics and Regenerative Medicine is the top non-industry sponsor for regenerative medicine clinical trials, followed by the University of Pennsylvania and Samsung Medical Center. Other leading non-industry sponsors are University of Miami, Mayo Clinic, and Standford University.
Regenerative Medicine Clinical Trials Analysis, by Top Non-Industry Sponsors, 2022 (%)
To know more about the leading non-industry sponsors of regenerative medicine clinical trials, download a free report sample.
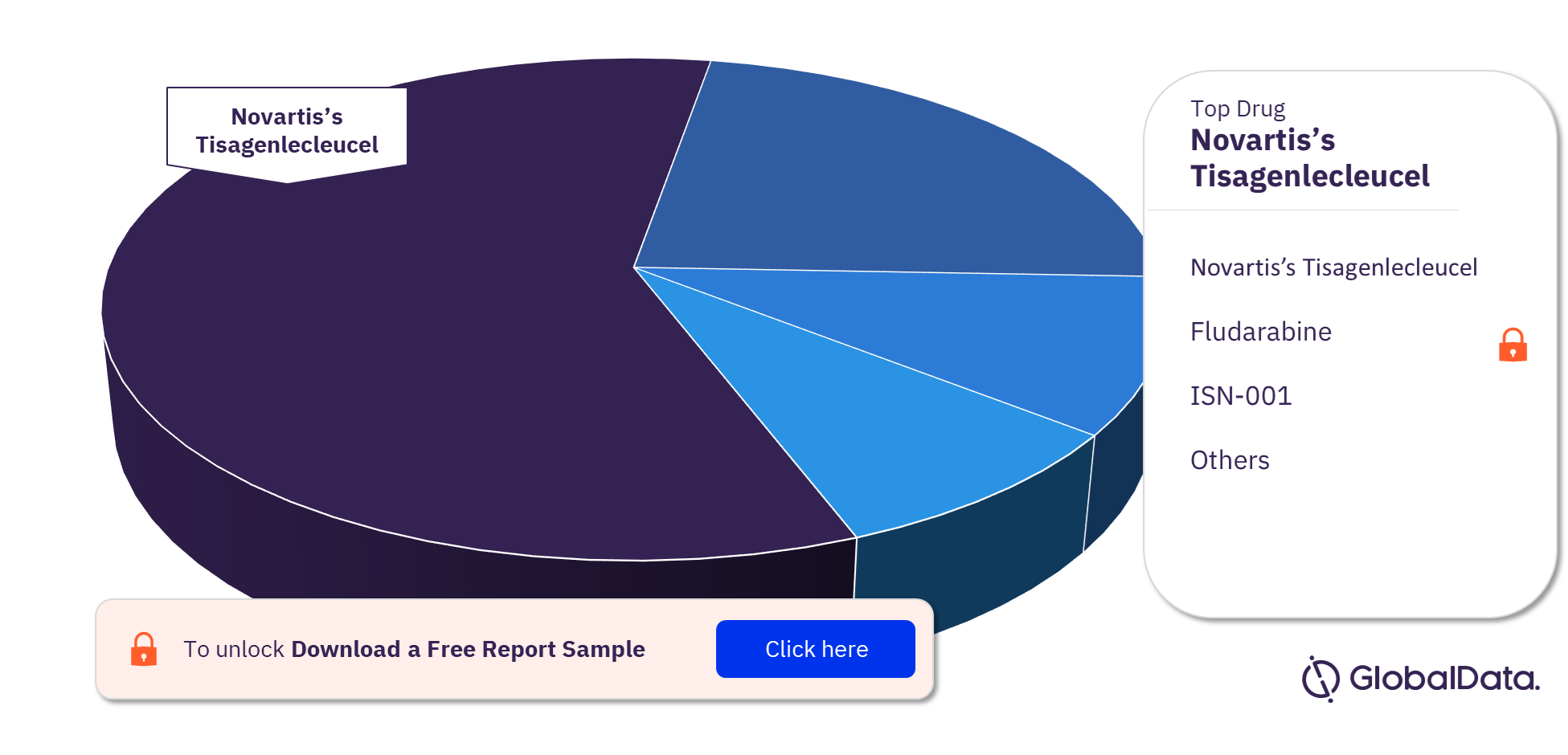
Regenerative Medicine Clinical Trials by Drugs
Novartis’s tisagenlecleucel, also known as Kymriah, is the top drug being tested in regenerative medicine trials. Other key drugs are Fludarabine, ISN-001, Stempeucel, and AlloRx Stem Cell Therapy among others.
Regenerative Medicine Clinical Trials Analysis by Top Drugs, 2022 (%)
To know more about the key drugs of regenerative medicine clinical trials, download a free report sample.
Scope
The clinical trials data used for these analyses were extracted from GlobalData’s Clinical Trials Database. The data was captured in the database as of September 8, 2022. The data has been analyzed by trial start year, phase, status, sponsor type, geography, single-country vs. multinational, virtual components, therapy area, indication, reasons for discontinuation, endpoint status, sponsors, and drugs.
Reasons to Buy
- Trends of regenerative medicine trials through the years
- Comparison of trials by sponsor type
- Can view top therapy areas, indications, geographies and sponsors
- Focus on the usage of decentralized clinical trials/virtual components
- Analysis of top drugs used
Table of Contents
Figures
Frequently asked questions
-
What are the key regions in the regenerative medicine clinical trials market?
North America, Asia-Pacific, Europe, South and Central America, and the Middle East and Africa are the key regions in the regenerative medicine clinical trials market.
-
What are the key therapy areas in the regenerative medicine clinical trials market?
cardiovascular, central nervous system, oncology, gastrointestinal, infectious disease, immunology, metabolic disorders, and ophthalmology are the key therapy areas in the regenerative medicine clinical trials market.
-
What are the key termination reasons in the regenerative medicine clinical trials market?
low accrual rate, business/strategic decisions, financial, product discontinuation, and lack of efficacy are the key termination reasons in the regenerative medicine clinical trials market.
-
Which are the leading industry sponsors in the regenerative medicine clinical trials market?
Mesoblast, Novartis, Anterogen, Pharmicell, Helixmith, and Anges are the leading industry sponsors in the regenerative medicine clinical trials market.
-
Which are the leading non-industry sponsors in the regenerative medicine clinical trials market?
The Foundation for Orthopaedics uand Regenerative Medicine, University of Pennsylvania, Samsung Medical Center, University of Miami, Mayo Clinic, And Standford University are the leading non-industry sponsors in the regenerative medicine clinical trials market.
-
What are the key drugs in the regenerative medicine clinical trials market?
Novartis’s Tisagenlecleucel, Fludarabine, ISN-001, Stempeucel, and Allorx Stem Cell Therapy are the key drugs in the regenerative medicine clinical trials market.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more Pharmaceuticals reports