Thrombocytopenia – Global Clinical Trials Review, H2, 2021
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
GlobalData’s clinical trial report provides data on the thrombocytopenia clinical trial scenario. This report provides top-line data relating to the clinical trials on Thrombocytopenia. The report includes an overview of trials count and their average enrolment in top countries conducted globally. The report includes coverage of disease clinical trials by region and top countries, trial phase, trial status, end points status, and sponsor type. The report also provides prominent drug names and trial counts for in-progress trials (based on the number of ongoing trials).
Thrombocytopenia clinical trial report consists of 1099 trials. Trials conducted by companies in Phase IV, Phase III, Phase II, Phase I, and Phase 0 stand at 84, 144, 176, 190, and 2 respectively. Similarly, the trials with different status compile of Completed – 683, Ongoing – 198, Planned – 72, Suspended – 3, Terminated – 77, and Withdrawn – 66. Out of 683 completed trials, 419 trials have achieved endpoints.
What are the market dynamics of the global thrombocytopenia therapeutics clinical trials sector?
The number of thrombocytopenia clinical trials conducted globally were increased by 19% for the period 2016-2020. The average number of patients enrolled was highest in the year 2019. As of November 2021, the majority of trials have been sponsored by institutions followed by companies and governments. Out of 683 completed trials, 480 trials have results and over 85% of trials reached endpoints. There were 198 clinical trials in progress and 683 trials were completed. The trials that were terminated/suspended or withdrawn accounted to 146. This was due to the lack of efficacy, safety, and lack of accrual of subjects. There were 198 clinical trials in progress of which is over 35% of trials were in Phase II study. There were 395 clinical trials in Phase II, of which 70 clinical trials were in progress for the time period 1995-2021.
What are the top regions and countries in the global thrombocytopenia therapeutics clinical trials sector?
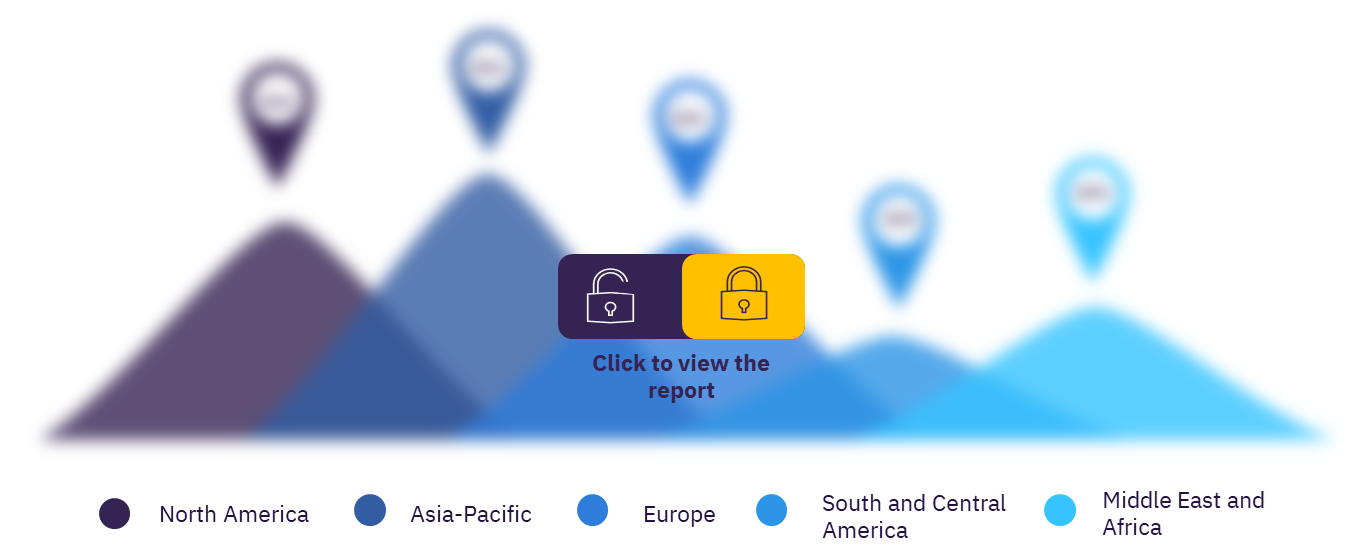
In total there were 1,099 clinical trials conducted on thrombocytopenia, as of November 2021, of these 490 clinical trials were in Asia-Pacific. More than 40% of the clinical trials were conducted in Asia-Pacific.
Asia-Pacific: As of November 2021, in the Asia-Pacific region, China has the highest number of thrombocytopenia clinical trials followed by Japan, India, Australia, and South Korea.
Europe: Among the European countries, Germany has the highest number of thrombocytopenia clinical trials, as of November 2021, followed by the UK, France, Italy, and Spain.
North America: In the country-wise analysis, the US has the highest number of thrombocytopenia clinical trials in North America followed by Canada and Mexico.
Middle East and Africa: Israel has the highest number of thrombocytopenia clinical trials followed by Iran, Egypt, Tunisia, and South Africa which are the leading countries in the region based on thrombocytopenia clinical trials.
Central and South America: Brazil has the highest number of thrombocytopenia clinical trials in the region followed by Argentina, Peru, Chile, and Colombia.
G7 countries: Among the G7 (the US, the UK, Germany, France, Italy, Canada, and Japan) countries, France has the highest proportion of thrombocytopenia to hematological disorders clinical trials. In total there were 822 clinical trials conducted on Thrombocytopenia, as of November 2021 in G7 Countries, of these 321 clinical trials were in the US. There were 91 clinical trials in progress and 317 trials are completed. The trials that are terminated/suspended or withdrawn accounted to 84. This was due to the lack of efficacy, safety, and lack of accrual of subjects.
E7 countries: Among the E7 (Brazil, Russia, India, China, Mexico, Turkey, and Indonesia) countries, China has the highest proportion of thrombocytopenia to hematological disorders clinical trials as of November 2021. In total there were 481 clinical trials conducted on Thrombocytopenia, as of November 2021 in E7 Countries, of these 294 clinical trials were in China. There were 103 clinical trials in progress and 219 trials were completed. The trials that were terminated/suspended or withdrawn accounted to 45. This was due to the lack of efficacy, safety, and lack of accrual of subjects.
Global thrombocytopenia therapeutics clinical trials sector, by regions
For more regional insights, download a free report sample
Which are the key companies in the global thrombocytopenia therapeutics clinical trials sector?
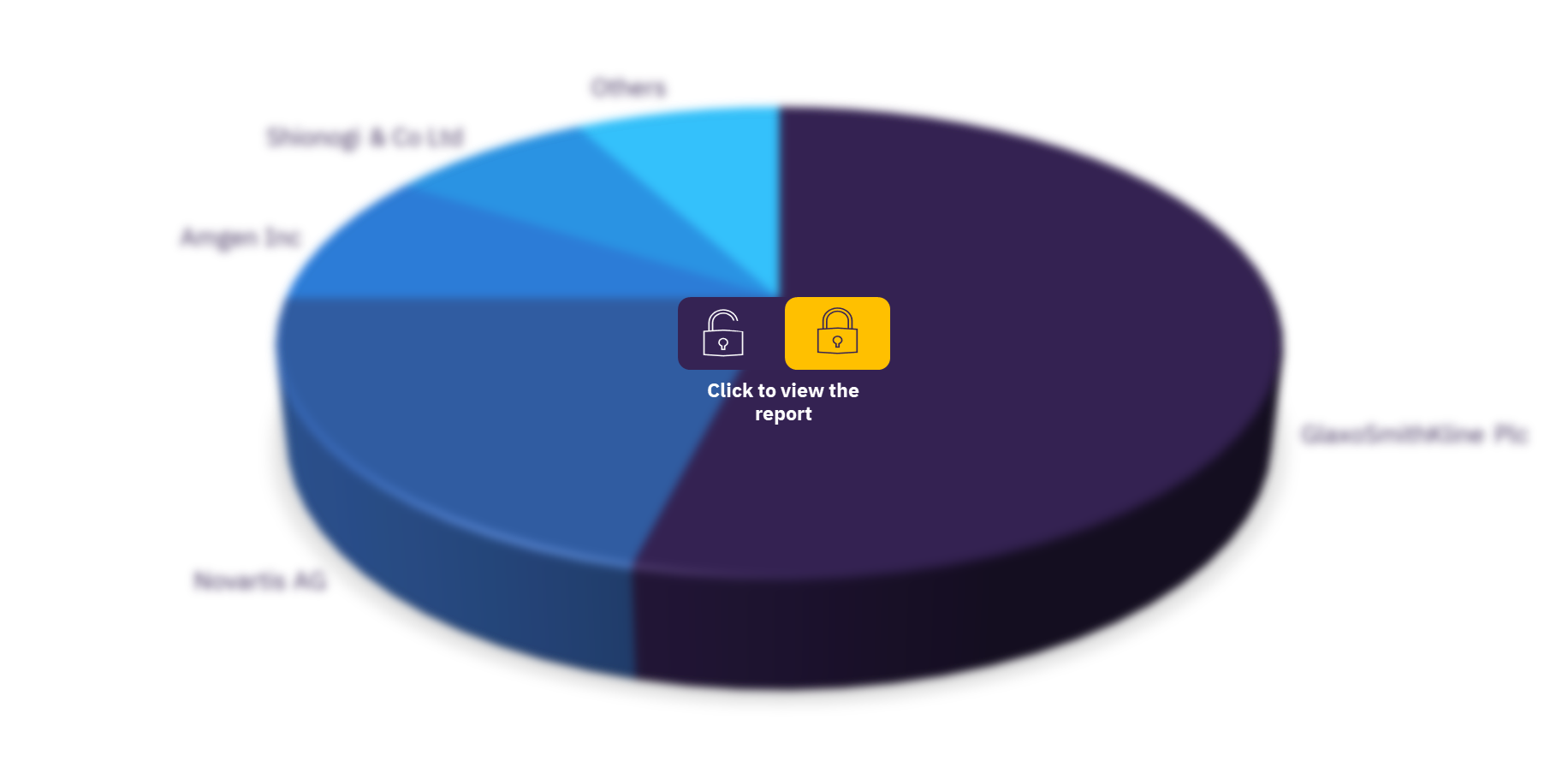
As of November 2021, GlaxoSmithKline Plc sponsored the highest number of thrombocytopenia clinical trials, followed by Novartis AG and Amgen Inc. Other major companies in the sector are Shionogi & Co Ltd, Swedish Orphan Biovitrum AB, Sanofi, Eisai Co Ltd, Rigel Pharmaceuticals Inc, 3SBio Inc, and F. Hoffmann-La Roche Ltd.
Global thrombocytopenia therapeutics clinical trials sector, by key companies
To know more about key companies, download a free report sample
Market report scope
| Key countries | The US, China, Germany, the UK, France, Italy, Spain, Japan, Russia, and Canada |
| Key companies | GlaxoSmithKline Plc, Novartis AG and Amgen Inc, Shionogi & Co Ltd, Swedish Orphan Biovitrum AB, Sanofi, Eisai Co Ltd, Rigel Pharmaceuticals Inc, 3SBio Inc, and F. Hoffmann-La Roche Ltd. |
Scope
- The report provides a snapshot of the global clinical trials landscape.
- Report provides top level data related to the clinical trials by Region, Country (G7 & E7), Trial Status, Trial Phase, Sponsor Type and End point status.
- The report reviews top companies involved and enlists all trials (Trial title, Phase, and Status) pertaining to the company.
- The report provides all the unaccomplished trials (Terminated, Suspended and Withdrawn) with reason for un-accomplishment.
- The Report provides enrollment trends for the past five years.
- Report provides latest news for the past three months.
Reasons to Buy
- Assists in formulating key business strategies with regards to investment.
- Helps in identifying prominent locations for conducting clinical trials which saves time and cost.
- Provides top level analysis of Global Clinical Trials Market which helps in identifying key business opportunities.
- Supports understanding of trials count and enrollment trends by country in the global therapeutics market.
- Aids in interpreting the success rates of clinical trials by providing a comparative scenario of completed and uncompleted (terminated, suspended or withdrawn) trials.
- Facilitates clinical trial assessment of the indication on a global, regional, and country-level.
Novartis AG
Amgen Inc
Shionogi & Co Ltd
Swedish Orphan Biovitrum AB
Sanofi
Eisai Co Ltd
Rigel Pharmaceuticals Inc
3SBio Inc
F. Hoffmann-La Roche Ltd
Table of Contents
Table
Figures
Frequently asked questions
-
What are the key countries in the global thrombocytopenia therapeutics clinical trials sector?
The key countries in the global thrombocytopenia therapeutics clinical trials sector are the US, China, Germany, the UK, France, Italy, Spain, Japan, Russia, and Canada.
-
Which are the key companies in the global thrombocytopenia therapeutics clinical trials sector?
The key companies in the global thrombocytopenia therapeutics clinical trials sector are GlaxoSmithKline Plc, Novartis AG and Amgen Inc, Shionogi & Co Ltd, Swedish Orphan Biovitrum AB, Sanofi, Eisai Co Ltd, Rigel Pharmaceuticals Inc, 3SBio Inc, and F. Hoffmann-La Roche Ltd.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more Hematological Disorders reports