Likelihood of Approval and Phase Transition Success Rate Model – Satralizumab in Demyelinating Diseases
Powered by ![]()
Unlock hidden opportunities in the LoA industry
Empower your strategies with our Likelihood of Approval and Phase Transition Success Rate Model – Satralizumab in Demyelinating Diseases report and make more profitable business decisions.
This report provides you with the data that allows you to track and predict the specific likelihood of approval (LOA) and phase transition success rate (PTSR) of a drug using GlobalData’s proprietary machine learning algorithms developed using over 10 years of historical data.
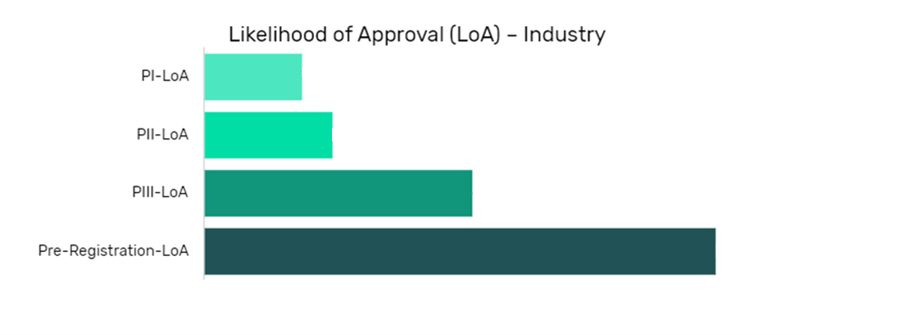
Satralizumab in Demyelinating Diseases Drug Details:
Satralizumab (Enspryng) is a humanized monoclonal antibody. It is formulated as solution for subcutaneous route of administration. Enspryng is indicated for the treatment of adult and children’s with neuromyelitis optica spectrum disorder (NMOSD) and uveitic macular edema. Enspryng is also indicated for the treatment of neuromyelitis optica spectrum disorder (NMOSD) in adult and adolescents patients who are anti-aquaporin-4 (AQP4) antibody positive.Satralizumab (SA-237) is under development for the treatment of neuromyelitis optica (NMO), myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD), Anti-N-Methyl-D-Aspartic Acid Receptor (NMDAR) Or Anti-Leucine-Rich Glioma-Inactivated 1 (LGI1) autoimmune encephalitis, duchenne muscular dystrophy, and NMO spectrum disorder and thyroid eye disease. It is administered subcutaneously as a solution. SA-237 is a humanized anti IL-6 receptor monoclonal antibody. It is a new molecular entity (NME). The drug candidate is developed from recycling antibody technology.The drug candidate was also under development for the treatment of aneurysmal subarachnoid hemorrhage, rheumatoid arthritis and myasthenia gravis.
Report Coverage
The data is segmented by drug name per indication and shows the current likelihood of approval for the drug compared to the indication benchmark and the industry benchmark.
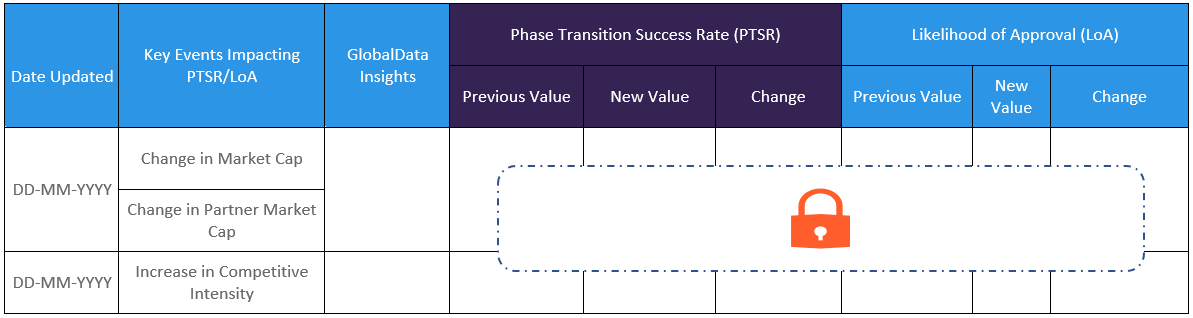
The Likelihood of Approval data is updated regularly based on events that take place which impact the clinical development process and regulatory considerations. GlobalData’s proprietary machine learning models consider these events in real time, to produce quantitative changes to the LOA and PTSR along with qualitative reasoning why the likelihood of approval has changed.
| Quick View – LOA Data | |||||
| Report Segments |
|
||||
| Drug Name |
|
||||
| Administration Pathway |
|
||||
| Therapeutic Areas | |||||
| Key Manufacturers |
|
||||
| Drug Development Status |
|
||||
Reasons to Buy
- Precise Likelihood of Approval and Phase Transition Success Rates: Our machine learning and proprietary models provide accurate predictions, helping you gauge the potential success of a drug in the regulatory process.
- Competitive Strategy Planning: Access information on LOA and PTSR for competitors’ drugs, allowing you to plan your clinical development, commercialisation and marketing strategies
- Event-driven Updates: Track event-driven changes in LOA and PTSR benchmarked against indication LOA/PTSR. Get the latest insights to adapt your strategies promptly!
- Well-informed Investment Decisions: This data helps you navigate the dynamic landscape of drug development and regulatory considerations.
Scope
- Drug Details: Drug name, Drug type, Intervention type
- Administration Pathway
- Therapeutic Areas
- Key Manufacturers
- Drug Development Status
This is an on-demand report that will be delivered upon request. The report will be delivered within 2 business days of the purchase, excluding weekends and holidays. Certain sections of the report may be removed or altered based on data availability and relevance.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Related reports
View more reports








