Likelihood of Approval and Phase Transition Success Rate Model – Finerenone in Congestive Heart Failure (Heart Failure)
Powered by ![]()
Unlock hidden opportunities in the LoA industry
Empower your strategies with our Likelihood of Approval and Phase Transition Success Rate Model – Finerenone in Congestive Heart Failure (Heart Failure) report and make more profitable business decisions.
This report provides you with the data that allows you to track and predict the specific likelihood of approval (LOA) and phase transition success rate (PTSR) of a drug using GlobalData’s proprietary machine learning algorithms developed using over 10 years of historical data.
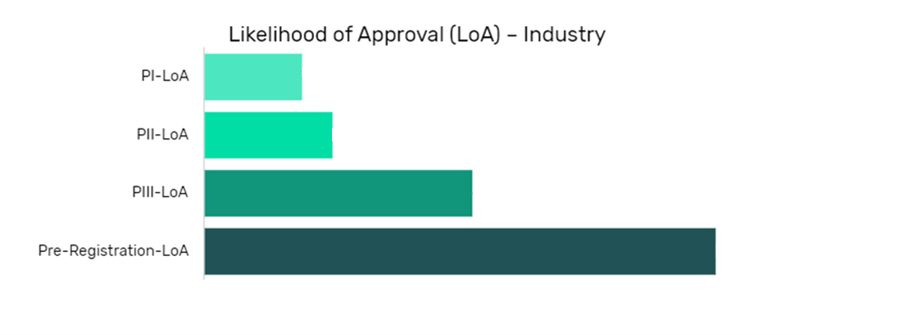
Finerenone in Congestive Heart Failure (Heart Failure) Drug Details:
Finerenone (Kerendia, Firialta) is a nonsteroidal mineralocorticoid. It is formulated as film coated tablets for oral route of administration. Kerendia is indicated to reduce the risk of sustained eGFR decline, end stage kidney disease, cardiovascular death, non-fatal myocardial infarction, and hospitalization for heart failure in adult patients with chronic kidney disease associated with type 2 diabetes. Kerendia is indicated for the treatment of chronic kidney disease (stage 3 and 4 with albuminuria) associated with type 2 diabetes in adults. Kerendia is indicated for the treatment of chronic kidney disease with type 2 diabetes in adults, excluding patients with end-stage renal disease or on dialysis. Kerendia is indicated for the treatment of chronic kidney disease (CKD) associated with type 2 diabetes (T2D) in adults, to reduce the risk of sustained eGFR decline and end-stage kidney disease.Finerenone (BAY-948862) is under development for the treatment of congestive heart failure and left ventricular ejection factor, non diabetic chronic kidney disease, proteinuria, glomerulonephritis and hypertension. The drug candidate is administered orally as a tablet. It is a new molecular entity and acts by targeting non-steroidal mineralocorticoid receptor (MR).It was under development for the treatment of Duchenne muscular dystrophy, diabetic retinopathy and diabetic nephropathy.
Report Coverage
The data is segmented by drug name per indication and shows the current likelihood of approval for the drug compared to the indication benchmark and the industry benchmark.
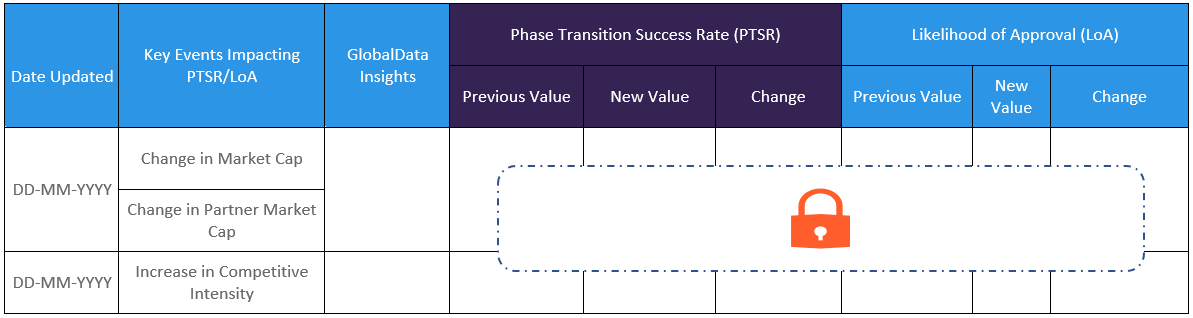
The Likelihood of Approval data is updated regularly based on events that take place which impact the clinical development process and regulatory considerations. GlobalData’s proprietary machine learning models consider these events in real time, to produce quantitative changes to the LOA and PTSR along with qualitative reasoning why the likelihood of approval has changed.
| Quick View – LOA Data | |||||
| Report Segments |
|
||||
| Drug Name |
|
||||
| Administration Pathway |
|
||||
| Therapeutic Areas | |||||
| Key Manufacturers |
|
||||
| Drug Development Status |
|
||||
Reasons to Buy
- Precise Likelihood of Approval and Phase Transition Success Rates: Our machine learning and proprietary models provide accurate predictions, helping you gauge the potential success of a drug in the regulatory process.
- Competitive Strategy Planning: Access information on LOA and PTSR for competitors’ drugs, allowing you to plan your clinical development, commercialisation and marketing strategies
- Event-driven Updates: Track event-driven changes in LOA and PTSR benchmarked against indication LOA/PTSR. Get the latest insights to adapt your strategies promptly!
- Well-informed Investment Decisions: This data helps you navigate the dynamic landscape of drug development and regulatory considerations.
Scope
- Drug Details: Drug name, Drug type, Intervention type
- Administration Pathway
- Therapeutic Areas
- Key Manufacturers
- Drug Development Status
This is an on-demand report that will be delivered upon request. The report will be delivered within 2 business days of the purchase, excluding weekends and holidays. Certain sections of the report may be removed or altered based on data availability and relevance.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Related reports
View more Pharmaceuticals reports









