03 Feb, 2021 COVID-19 disrupted clinical trials due to slow recruitment continue to grow
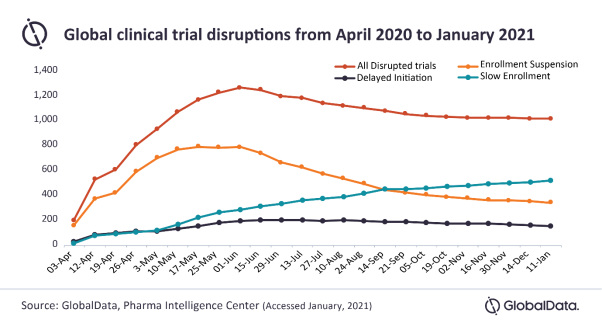
Posted in CoronavirusSince June 2020, the number of total disrupted trials has been falling slowly; however, this has leveled off in October 2020. Despite this, the number of clinical trials that have resumed continues to rise. The majority of current trial disruptions are due to trials impacted by slow enrollment, which continue to gradually increase, says GlobalData, a leading data and analytics company.
Brooke Wilson, Associate Director, Trials Intelligence at GlobalData, comments: “This implies that although sponsors and contract service providers have begun to adjust clinical trial design strategies and are adapting to the new post-COVID-19 environment, the surge of the new variant and subsequent cases is having an impact.”
Trials impacted by enrollment suspension have been on a downward trajectory while clinical trials that delayed initiation have remained steady. This suggests trials that had initiated enrollment before the pandemic with chosen sites and investigators, but then suspended due to COVID-19, are having more success picking up where they left off when enrollment was not impacted.
Within the category of trials currently affected by slow enrollment, 7.2% of these are specifically due to the unavailability of sites and investigators.
Wilson continues: “Many hospitals that serve as trial sites were inundated with COVID-19 patients and may still not be available especially due to the emergence of a new COVID-19 variant and spike in confirmed cases, likewise, many investigators may have been reassigned to COVID-19 drug discovery trials or treating COVID-19 patients and the activation of sites for non-COVID-19 trials is being deprioritized.”
As the number of trials that have been impacted by slow enrollment continues to increase, this continues to be an issue.
Wilson adds: “There is a high risk to subjects in a clinical trial who have a serious chronic or acute condition that affects their immune system, giving them a greater chance of contracting COVID-19 and making them unwilling to enroll in a clinical trial.”
