Interleukin 1 Receptor Associated Kinase 4 (Renal Carcinoma Antigen NY REN 64 or IRAK4 or EC 2.7.11.1) Drugs in Development by Stages, Target, MoA, RoA, Molecule Type and Key Players, 2022 Update
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
IRAK-4 Drugs in Development Report Overview
Interleukin-1 receptor-associated kinase 4 (IRAK-4) is a protein kinase involved in signaling innate immune responses from Toll-like receptors. The Interleukin 1 Receptor Associated Kinase 4 (IRAK-4) drugs in development research report provides comprehensive information on the therapeutics under development for IRAK-4. The report also analyzes the stage of development, mechanism of action (MoA), route of administration (RoA), molecule type, therapy area, and indication.
The pipeline report covers the descriptive pharmacological action and product description of IRAK-4. It includes the complete research and development history as well as the latest news and press releases. Additionally, the report provides an overview of key players involved in IRAK-4 therapeutic development and features dormant and discontinued products.
Buy the Full Report to Know More about the IRAK-4 Pipeline Products
| Key Therapy Areas | · Immunology
· Oncology · Dermatology · Gastrointestinal · Metabolic Disorders |
| Key Indications | · Rheumatoid Arthritis
· Inflammation · Atopic Dermatitis (Atopic Eczema) · Myelodysplastic Syndrome · Autoimmune Disorders |
| Key Mechanism of Action | · Interleukin 1 Receptor Associated Kinase 4 Inhibitor |
| Key Route of Administration | · Oral
· Topical · Intravenous |
| Key Molecule Type | · Small Molecule |
| Leading Companies | · Kymera Therapeutics Inc
· Rigel Pharmaceuticals Inc · Asahi Kasei Pharma Corp · Astellas Pharma Inc · AstraZeneca Plc |
| Enquiry & Decide | Discover the perfect solution for your business needs. Enquire now and let us help you make an informed decision before making a purchase. |
Interleukin 1 Receptor Associated Kinase 4 Pipeline Products Segmentation by Therapy Areas
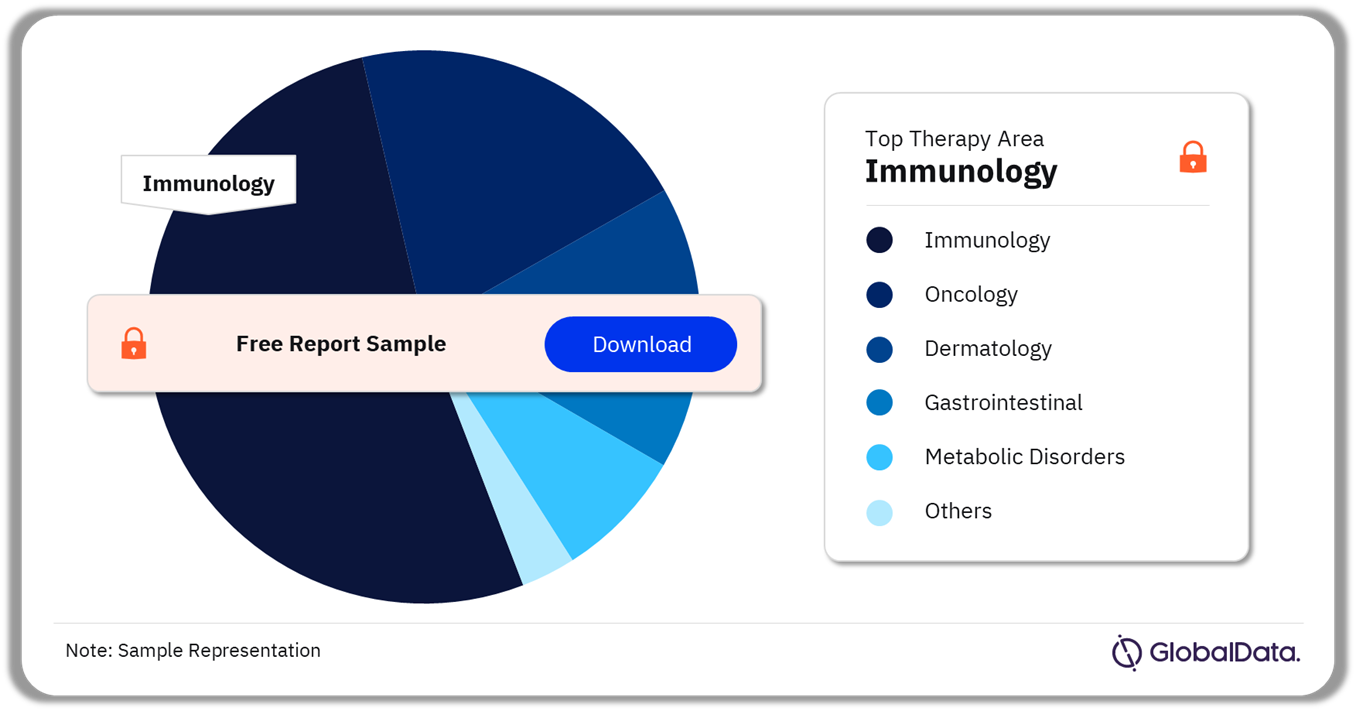
The key therapy areas associated with IRAK-4 drugs in development include immunology, oncology, dermatology, gastrointestinal, and metabolic disorders, among others. Immunology had the highest share among IRAK-4 drugs in development in 2022.
IRAK-4 Pipeline Products Analysis by Therapy Areas, 2022 (%)
Buy the Full Report for More Therapy Area Insights into the IRAK-4 Pipeline Products
Interleukin 1 Receptor Associated Kinase 4 Pipeline Products Segmentation by Indications
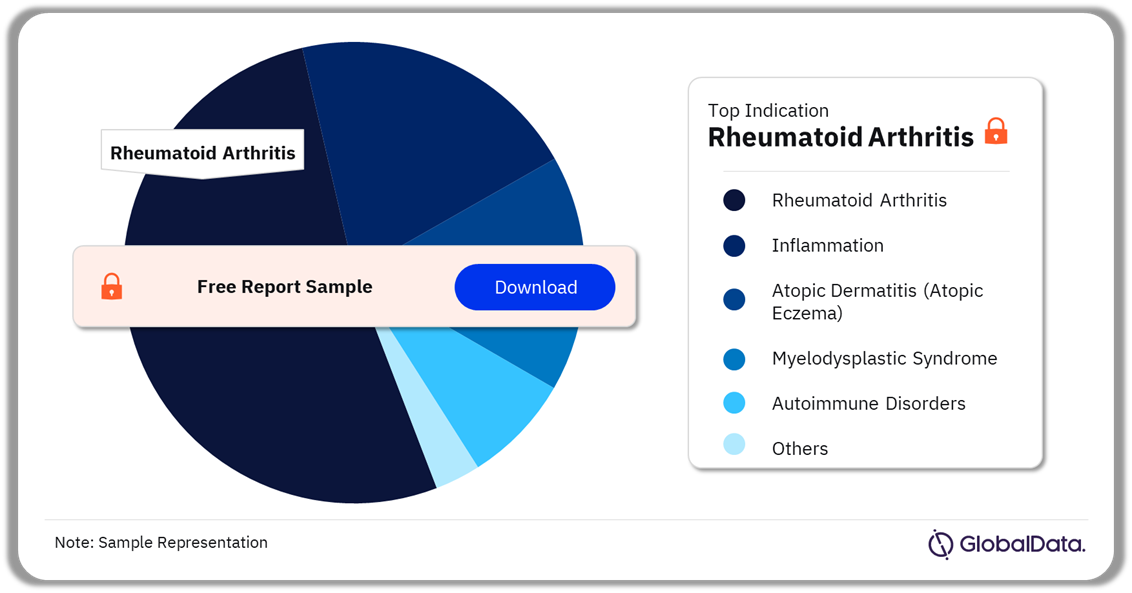
A few of the key indications for IRAK-4 drug application include rheumatoid arthritis, inflammation, atopic dermatitis (atopic eczema), myelodysplastic syndrome, and autoimmune disorders, among others. IRAK-4 drugs had the highest number of products in development for rheumatoid arthritis in 2022.
IRAK-4 Pipeline Products Analysis by Indications, 2022 (%)
Buy the Full Report for More Indication Insights into IRAK-4 Pipeline Products
Interleukin 1 Receptor Associated Kinase 4 Pipeline Products Segmentation by MoA, RoA, and Molecule Types
The mechanism of action in IRAK-4 drugs in development is Interleukin 1 Receptor Associated Kinase 4 Inhibitor and the key molecule type is the small molecule.
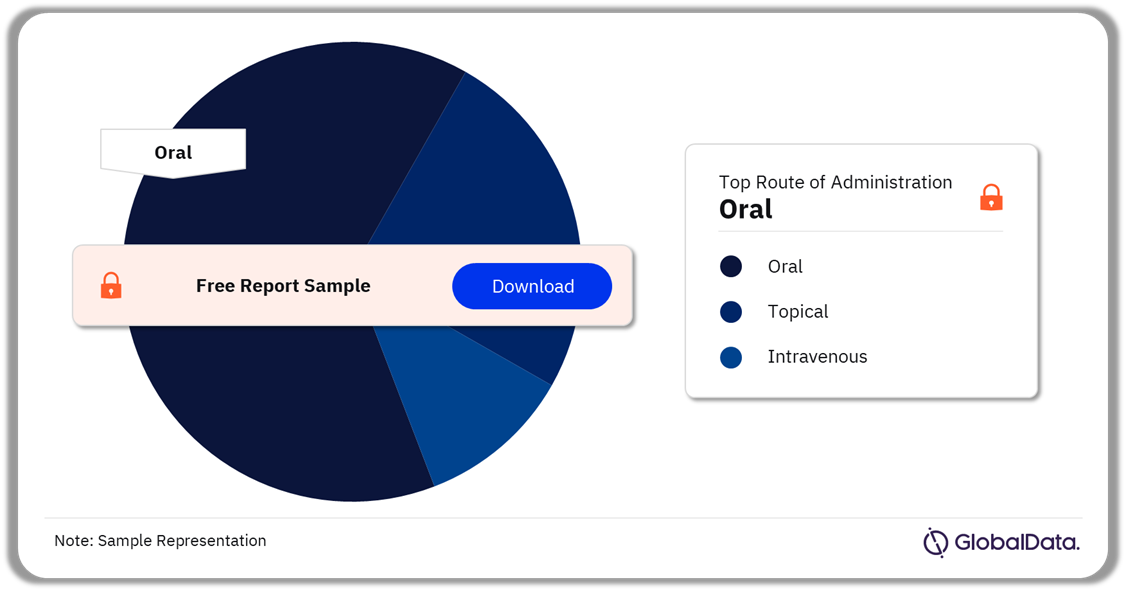
The IRAK-4 drugs can be administered orally, topically, or through the veins. Oral RoA was the most preferred way for IRAK-4 drug administration in 2022.
IRAK-4 Pipeline Products Analysis by Routes of Administration, 2022 (%)
Buy the Full Report for More MoA, RoA, and Molecule Type Insights into IRAK-4 Pipeline Products
Interleukin 1 Receptor Associated Kinase 4 Pipeline Products - Competitive Landscape
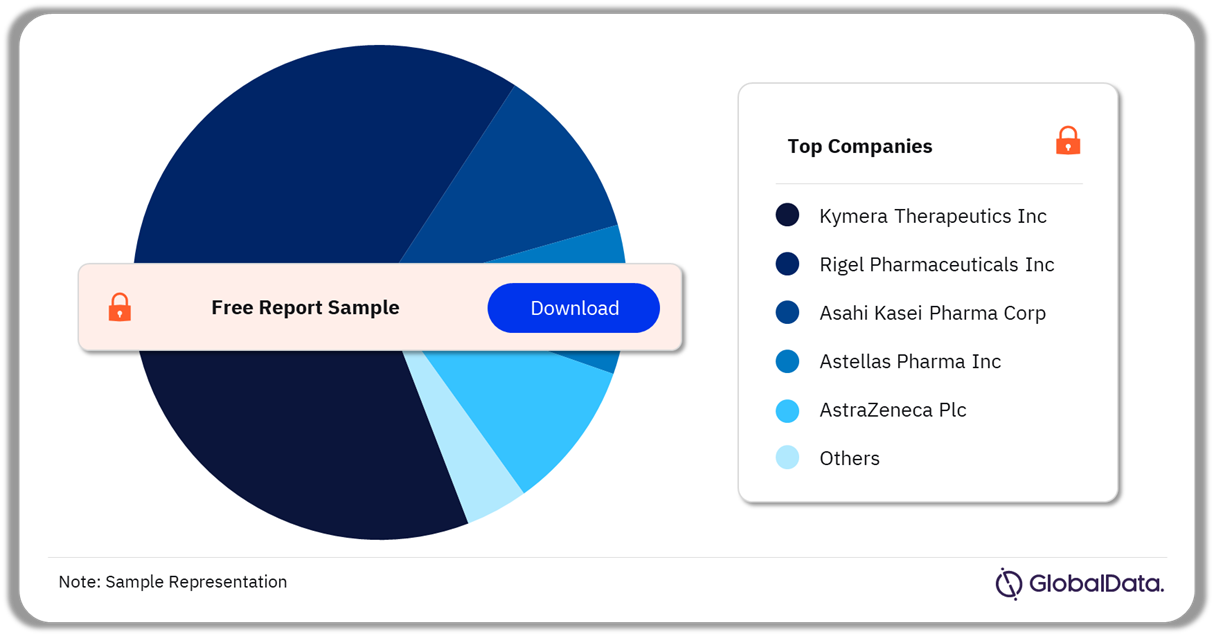
Kymera Therapeutics Inc. and Rigel Pharmaceuticals Inc. had the highest number of products in the IRAK-4 pipeline in 2022
A few of the leading companies associated with IRAK-4 pipeline products are:
- Kymera Therapeutics Inc
- Rigel Pharmaceuticals Inc
- Asahi Kasei Pharma Corp
- Astellas Pharma Inc
- AstraZeneca Plc
Kymera Therapeutics Inc.: Kymera has headquarters in Watertown, Massachusetts, the US. It is a clinical-stage biopharma and technology company involved in developing small molecule therapeutics for protein degradation. The company also carries out research and discovery of breakthrough drugs addressing novel medical areas. Its products find application in cancers, inflammatory, autoimmune, and other related diseases.
IRAK-4 Pipeline Products Analysis by Companies, 2022 (%)
Buy the Full Report for More Company Insights into IRAK-4 Pipeline Products
Interleukin 1 Receptor Associated Kinase 4 Pipeline Products - Featured News
16th December 2022: Rigel Pharmaceuticals has dosed the first subject in its Phase Ib clinical trial of investigational therapy R289 to treat lower-risk myelodysplastic syndrome (MDS) patients who were refractory or resistant to previous therapies. The safety of R289 is its primary objective. Evaluating the initial efficacy and characterizing the pharmacokinetic (PK) and pharmacodynamic (PD) profile of the therapy are the trial’s secondary and exploratory objectives.
Buy the Full Report to Know More Featured News about IRAK-4 Pipeline Products
Segments Covered in the Report
IRAK-4 Pipeline Products Therapy Area Outlook (2022)
- Immunology
- Oncology
- Dermatology
- Gastrointestinal
- Metabolic Disorders
IRAK-4 Pipeline Products Indication Outlook (2022)
- Rheumatoid Arthritis
- Inflammation
- Atopic Dermatitis (Atopic Eczema)
- Myelodysplastic Syndrome
- Autoimmune Disorders
IRAK-4 Pipeline Products MoA Outlook (2022)
- Interleukin 1 Receptor Associated Kinase 4 Inhibitor
IRAK-4 Pipeline Products RoA Outlook (2022)
- Oral
- Topical
- Intravenous
IRAK-4 Pipeline Products Molecule Type Outlook (2022)
- Small molecule
Scope
The report provides:
- A snapshot of the global therapeutic landscape of Interleukin 1 Receptor Associated Kinase 4.
- Review of pipeline therapeutics for Interleukin 1 Receptor Associated Kinase 4 by companies and universities/research institutes. The review is based on information derived from company and industry-specific sources.
- Information about pipeline products based on several stages of development ranging from pre-registration to discovery and undisclosed stages.
- Descriptive drug profiles for the pipeline products which comprise product description, descriptive licensing and collaboration details, R&D brief, MoA & other developmental activities.
- Key companies involved in Interleukin 1 Receptor Associated Kinase 4 therapeutics and enlist all their major and minor products for the indication.
- Evaluation of Interleukin 1 Receptor Associated Kinase 4 therapeutics based on their mechanism of action (MoA), route of administration (RoA), and molecule type.
- All the dormant and discontinued pipeline products.
- Reviews of the latest news related to pipeline therapeutics for Interleukin 1 Receptor Associated Kinase 4.
Reasons to Buy
- Procure strategically important competitor information, analysis, and insights to formulate effective R&D strategies.
- Recognize emerging players with potentially strong product portfolios and create effective counterstrategies to gain competitive advantage.
- Find and recognize significant and varied types of therapeutics under development for Interleukin 1 Receptor Associated Kinase 4.
- Classify potential new clients or partners in the target demographic.
- Develop tactical initiatives by understanding the focus areas of leading companies in the Interleukin 1 Receptor Associated Kinase 4 market.
- Plan mergers and acquisitions meritoriously by identifying key players and their most promising pipeline therapeutics.
- Formulate corrective measures for pipeline projects by understanding the Interleukin 1 Receptor Associated Kinase 4 pipeline depth and the focus of therapeutics.
- Develop and design in-licensing and out-licensing strategies by identifying prospective partners with the most attractive products to enhance and expand business potential and scope.
- Adjust the therapeutic portfolio by recognizing discontinued products and applying this knowledge to pipeline development.
Astellas Pharma Inc
AstraZeneca Plc
Aurigene Discovery Technologies Ltd
Bayer AG
Beijing Hanmi Pharmaceutical Co Ltd
Bristol-Myers Squibb Co
Chia Tai Tianqing Pharmaceutical Group Co Ltd
Emmaus Life Sciences Inc
Evommune Inc
Fresh Tracks Therapeutics Inc
Gilead Sciences Inc
Kurome Therapeutics Inc
Kymera Therapeutics Inc
Nyrada Inc
Pfizer Inc
Rigel Pharmaceuticals Inc
TG Therapeutics Inc
Xi'An Yufan Biotechnology Co Ltd
Table of Contents
Table
Figures
Frequently asked questions
-
Which was the leading therapy area in IRAK-4 drugs in development in 2022?
Immunology had the highest share among IRAK-4 drugs in development in 2022.
-
Which was the leading indication for IRAK-4 drugs in development in 2022?
IRAK-4 drugs had the highest number of products in development for rheumatoid arthritis in 2022.
-
Which was the leading route of administration for IRAK-4 drugs in development in 2022?
Oral RoA was the most preferred way for IRAK-4 drug administration in 2022.
-
Which are the leading companies associated with IRAK-4 drugs in development?
A few of the leading companies associated with the IRAK-4 pipeline products are Kymera Therapeutics Inc, Rigel Pharmaceuticals Inc, Asahi Kasei Pharma Corp, Astellas Pharma Inc, and AstraZeneca Plc, among others.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more Other Diseases reports