Likelihood of Approval and Phase Transition Success Rate Model – Disitamab Vedotin in Kidney Cancer (Renal Cell Cancer)
Powered by ![]()
Unlock hidden opportunities in the LoA industry
Empower your strategies with our Likelihood of Approval and Phase Transition Success Rate Model – Disitamab Vedotin in Kidney Cancer (Renal Cell Cancer) report and make more profitable business decisions.
This report provides you with the data that allows you to track and predict the specific likelihood of approval (LOA) and phase transition success rate (PTSR) of a drug using GlobalData’s proprietary machine learning algorithms developed using over 10 years of historical data.
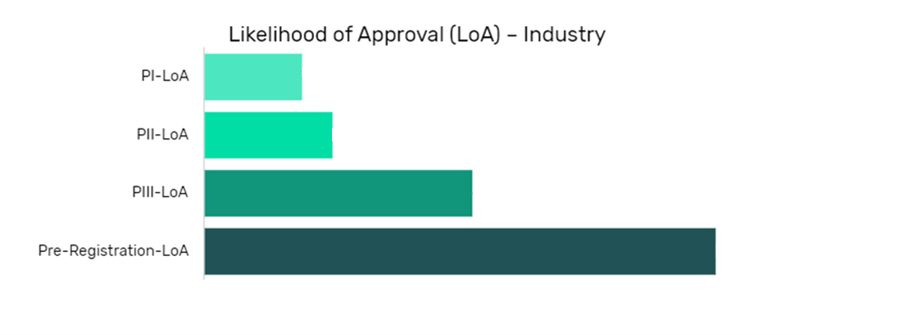
Disitamab Vedotin in Kidney Cancer (Renal Cell Cancer) Drug Details:
Disitamab vedotin (Aidexi) is an antibody-conjugated drug that contains the human epidermal growth factor receptor-2 (HER2) antibody portion, linker and the cytotoxic drug monomethyl auristatin E (MMAE). It is developed based on antibody-drug conjugates technology. It is formulated as solution for injection for intravenous route of administration. Aidexi is indicated for the treatment of locally advanced or metastatic gastric cancer (including gastroesophageal junction adenocarcinoma) with HER2 over-expression in patients who have received at least two types of systemic chemotherapy.Disitamab vedotin (RC-48) is under development for the treatment of solid tumors including breast cancer, HER2 expressing non-small cell lung cancer, non-muscle invasive bladder cancer (NMIBC), metastatic liver cancer, advanced or metastatic urothelial carcinoma of unresectable origin including urothelial carcinoma of bladder, ureter cancer, renal pelvis cancer, and urethral cancer, advanced melanoma, advanced or metastatic HER2 expressing biliary tract cancer, bile duct cancer, gallbladder cancer, muscle invasive bladder cancer (MIBC), HER2 expressing urothelial carcinoma and HER2 low to non expressing urothelial cancer, metastatic cholangiocarcinoma, HER2 expressing gastric cancer, gastro-esophageal junction cancer or gastroesophageal junction adenocarcinoma and refractory HER2 low expressing metastatic breast cancer, HER2 positive and negative breast cancers, cervical cancer, epithelial ovarian cancer, fallopian tube cancer, peritoneal cancer, endometrial cancer, vulvar cancer, vaginal cancer, metastatic colorectal cancer, salivary gland tumor, head and neck squamous cell carcinoma, esophageal carcinoma, metastatic castration resistant prostate cancer (mCRPC), oral cavity cancer, oropharyngeal cancer, hypopharyngeal cancer, laryngeal cancer and primary sarcoma of the gynecological reproductive system. It is a recombinant humanized anti-HER2 monoclonal antibody-monomethyl auristatin E (MMAE) antibody-drug conjugate administered through intravenous and intravenous drip routes in the form of powder for solution. The drug candidate acts by targeting the human epidermal growth factor receptor 2 (Her2). The drug candidate is developed based on antibody-drug conjugates technology. It was also under development for the treatment of ovarian cancer and bladder cancer
Report Coverage
The data is segmented by drug name per indication and shows the current likelihood of approval for the drug compared to the indication benchmark and the industry benchmark.
The Likelihood of Approval data is updated regularly based on events that take place which impact the clinical development process and regulatory considerations. GlobalData’s proprietary machine learning models consider these events in real time, to produce quantitative changes to the LOA and PTSR along with qualitative reasoning why the likelihood of approval has changed.
| Quick View – Disitamab Vedotin LOA Data | |||||
| Report Segments |
|
||||
| Drug Name |
|
||||
| Administration Pathway |
|
||||
| Therapeutic Areas |
|
||||
| Key Manufacturers |
|
||||
| Drug Development Status |
|
||||
Reasons to Buy
- Precise Likelihood of Approval and Phase Transition Success Rates: Our machine learning and proprietary models provide accurate predictions, helping you gauge the potential success of a drug in the regulatory process.
- Competitive Strategy Planning: Access information on LOA and PTSR for competitors’ drugs, allowing you to plan your clinical development, commercialisation and marketing strategies
- Event-driven Updates: Track event-driven changes in LOA and PTSR benchmarked against indication LOA/PTSR. Get the latest insights to adapt your strategies promptly!
- Well-informed Investment Decisions: This data helps you navigate the dynamic landscape of drug development and regulatory considerations.
Scope
- Drug Details: Drug name, Drug type, Intervention type
- Administration Pathway
- Therapeutic Areas
- Key Manufacturers
- Drug Development Status
This is an on-demand report that will be delivered upon request. The report will be delivered within 2 business days of the purchase, excluding weekends and holidays. Certain sections of the report may be removed or altered based on data availability and relevance.
Frequently asked questions
- Drugs which have been approved in the past 10 years
- Drugs which have failed during clinical development in the past 18 years
- Drugs which are currently in development
- Phase I, Phase II, Phase III, and Pre-Registration development stage
- Drugs must meet one of the following criteria to be included in the model:
- The developer has specified the US as an intended market for approval.
- The developer has not specified any country as an intended market for approval, i.e. the “Drug Geography” is listed as “Global”
- Innovator drugs and biosimilars
- Diagnostics, Imaging Agents, Biomarkers, stents and other drug delivery devices (covered in GlobalData’s Medical Intelligence Center).
- Nutraceuticals, dietary supplements, alternative medicines, imaging agents, radio emitter, transplants, transfusions, fillers, cosmetics, probiotics, antiseptics, antacids, mobilizing agents, veterinary drugs and drugs not seeking approval.
- Generic drugs
- Innovative drugs in Preclinical or Discovery Stage.
- Pipeline drugs sponsored by a Government or Institution.
- Drugs with a specific Drug Geography not the United States.
The probability of a drug ultimately receiving market authorization
The probability of a drug’s advancement to the next stage of clinical development
GlobalData’s Drug-Specific Likelihood of Approval (LoA) calculates the Phase Transition Success Rate (PTSR) and Likelihood of Approval (LoA) customized to individual drug. The model uses a combination of Machine Learning (ML) and a GlobalData proprietary algorithm to process data points from the Drugs, Clinical Trials, Regulatory Milestones, Company, and Financial databases.
Inclusion
Data Scope:
Drug Phase Scope:
Drug Geography Scope:
Drug Type Scope:
Entity Type Scope:
Only drugs in development by companies are included in the model.
Exclusion
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Related reports
View more Pharmaceuticals reports









