Epilepsy Drugs in Development by Stages, Target, MoA, RoA, Molecule Type and Key Players, 2022 Update
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
Epilepsy is a neurological disorder characterized by the transmission of abnormal signals produced by a group of neurons in the brain which causes seizures. This leads to brief disruptions of the senses and short periods of unconsciousness or convulsions. Symptoms of epilepsy are repeated seizures, weakness, anxiety, loss of consciousness, and contraction or jerking of body muscles.
The Epilepsy Drugs in Development market research report provides an overview of the Epilepsy pipeline landscape. The report provides comprehensive information on the therapeutics under development for Epilepsy, complete with analysis by stage of development, drug target, mechanism of action (MoA), route of administration (RoA), and molecule type. The report also covers the descriptive pharmacological action of the therapeutics, its complete research and development history and latest news and press releases. Additionally, the report provides an overview of key players involved in therapeutic development for Epilepsy and features dormant and discontinued projects.
Epilepsy Pipeline Drugs Market Targets
The targets of the epilepsy pipeline drugs market are Gamma-Aminobutyric Acid Type A Receptor Subunit (GABA, Voltage Gated Sodium Channel, Cannabinoid Receptor 2, Cannabinoid Receptor 1, Potassium Voltage Gated Channel Subfamily KQT Member 2, Potassium Voltage Gated Channel Subfamily KQT Member 3, Sodium Channel Protein Type 8 Subunit Alpha, Sodium Channel Protein Type 1 Subunit Alpha, 5-Hydroxytryptamine Receptor, and Serine/Threonine Protein Kinase mTOR.
Epilepsy pipeline drugs market, by targets
For more target insights, download a free report sample
Mechanisms of Action of Epilepsy Pipeline Drugs Market
The mechanisms of action of the epilepsy pipeline drugs market are Gamma-Aminobutyric Acid Type A Receptor Subunit (GABAAgonist, Voltage Gated Sodium Channel Blocker, Cannabinoid Receptor 2 Agonist, Cannabinoid Receptor 1 Agonist, Potassium Voltage Gated Channel Subfamily KQT Member 2 Activator, Potassium Voltage Gated Channel Subfamily KQT Member 3 Activator, Sodium Channel Protein Type 1 Subunit Alpha Activator, Sodium Channel Protein Type 8 Subunit Alpha Blocker, 5-Hydroxytryptamine Receptor Agonist, and Serine/Threonine Protein Kinase mTOR Inhibitor.
Epilepsy pipeline drugs market, by mechanisms of action
For more mechanisms of action insights, download a free report sample
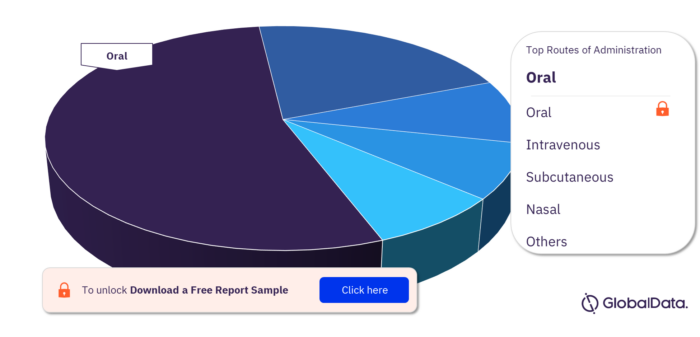
Routes of Administration in Epilepsy Pipeline Drugs Market
The routes of administration in the epilepsy pipeline drugs market are oral, intravenous, subcutaneous, nasal, intrathecal, buccal, inhalational, sublingual, transdermal, and parenteral.
Epilepsy pipeline drugs market, by routes of administration
For more routes of administration insights, download a free report sample
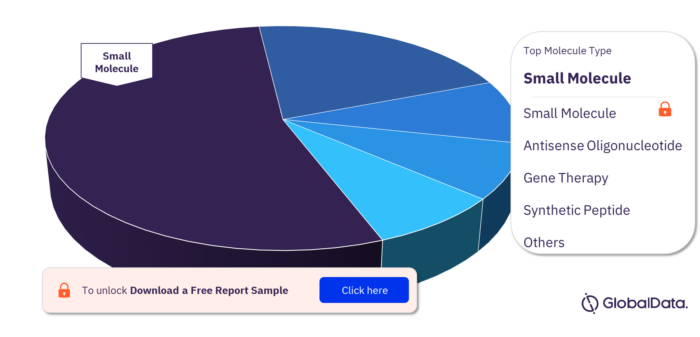
Molecule Types in Epilepsy Pipeline Drugs Market
The molecule types in the epilepsy pipeline drugs market are small molecule, antisense oligonucleotide, gene therapy, synthetic peptide, cell therapy, RNAi gene therapy, antibody, antisense RNAi oligonucleotide, biologic, and fusion protein.
Epilepsy pipeline drugs market, by molecule types
For more molecule type insights, download a free report sample
Key Epilepsy Pipeline Drugs Market Companies
Some of the key companies in the epilepsy pipeline drugs market are Praxis Precision Medicines Inc, EpyGenix Therapeutics Inc, Neurocrine Biosciences Inc, Bio-Pharm Solutions Co Ltd, Q-State Biosciences Inc, Taysha Gene Therapies Inc, UCB SA, Jazz Pharmaceuticals Plc, SK Biopharmaceuticals Co Ltd, and AfaSci Inc.
Epilepsy pipeline drugs market, by key companies
To know more about key companies, download a free report sample
Market report overview
| Targets | Gamma-Aminobutyric Acid Type A Receptor Subunit (GABA, Voltage Gated Sodium Channel, Cannabinoid Receptor 2, Cannabinoid Receptor 1, Potassium Voltage Gated Channel Subfamily KQT Member 2, Potassium Voltage Gated Channel Subfamily KQT Member 3, Sodium Channel Protein Type 8 Subunit Alpha, Sodium Channel Protein Type 1 Subunit Alpha, 5-Hydroxytryptamine Receptor, and Serine/Threonine Protein Kinase mTOR |
| Mechanisms of Action | Gamma-Aminobutyric Acid Type A Receptor Subunit (GABAAgonist, Voltage Gated Sodium Channel Blocker, Cannabinoid Receptor 2 Agonist, Cannabinoid Receptor 1 Agonist, Potassium Voltage Gated Channel Subfamily KQT Member 2 Activator, Potassium Voltage Gated Channel Subfamily KQT Member 3 Activator, Sodium Channel Protein Type 1 Subunit Alpha Activator, Sodium Channel Protein Type 8 Subunit Alpha Blocker, 5-Hydroxytryptamine Receptor Agonist, and Serine/Threonine Protein Kinase mTOR Inhibitor |
| Routes of Administration | Oral, Intravenous, Subcutaneous, Nasal, Intrathecal, Buccal, Inhalational, Sublingual, Transdermal, and Parenteral |
| Molecule Types | Small Molecule, Antisense Oligonucleotide, Gene Therapy, Synthetic Peptide, Cell Therapy, RNAi Gene Therapy, Antibody, Antisense RNAi Oligonucleotide, Biologic, and Fusion Protein |
| Key Companies | Praxis Precision Medicines Inc, EpyGenix Therapeutics Inc, Neurocrine Biosciences Inc, Bio-Pharm Solutions Co Ltd, Q-State Biosciences Inc, Taysha Gene Therapies Inc, UCB SA, Jazz Pharmaceuticals Plc, SK Biopharmaceuticals Co Ltd, and AfaSci Inc |
Scope
This report provides:
- A snapshot of the global therapeutic landscape of Epilepsy (Central Nervous System).
- Reviews of pipeline therapeutics for Epilepsy (Central Nervous System) by companies and universities/research institutes based on information derived from company and industry-specific sources.
- Pipeline products based on various stages of development ranging from pre-registration to discovery and undisclosed stages.
- Descriptive drug profiles for the pipeline products which include product description, descriptive MoA, R&D brief, licensing and collaboration details & other developmental activities.
- Reviews of key companies involved in Epilepsy (Central Nervous System) therapeutics and enlists all their major and minor projects.
- Evaluation of Epilepsy (Central Nervous System) targeted therapeutics based on drug target, mechanism of action (MoA), route of administration (RoA), and molecule type.
- All the dormant and discontinued pipeline projects.
- Reviews of the latest news and deals related to pipeline therapeutics for Epilepsy (Central Nervous System).
Reasons to Buy
- Procure strategically important competitor information, analysis, and insights to formulate effective R&D strategies.
- Recognize emerging players with potentially strong product portfolio and create effective counter-strategies to gain competitive advantage.
- Find and recognize significant and varied types of therapeutics under development for Epilepsy (Central Nervous System).
- Classify potential new clients or partners in the target demographic.
- Develop tactical initiatives by understanding the focus areas of leading companies.
- Plan mergers and acquisitions meritoriously by identifying key players and it’s most promising pipeline therapeutics.
- Formulate corrective measures for pipeline projects by understanding Epilepsy (Central Nervous System) pipeline depth and focus of Indication therapeutics.
- Develop and design in-licensing and out-licensing strategies by identifying prospective partners with the most attractive projects to enhance and expand business potential and scope.
- Adjust the therapeutic portfolio by recognizing discontinued projects and understand from the know-how what drove them from pipeline.
Aadi Bioscience Inc
Accure Therapeutics SL
Advanced Neural Dynamics Inc
Advicenne SA
Aequus Pharmaceuticals Inc
AfaSci Inc
Affectis Pharmaceuticals AG
AgoneX Biopharmaceuticals Inc
Altamira Therapeutics Ltd
AMO Pharma Ltd
Anavex Life Sciences Corp
Aphios Corp
Apteeus SAS
Aquestive Therapeutics Inc
Arovella Therapeutics Ltd
Aucta Pharmaceuticals LLC
Aurum Biosciences Ltd
Avicanna Inc
Azurity Pharmaceuticals Inc
B&A Therapeutics
Baergic Bio Inc
BCWorld Pharm Co Ltd
Bio-Pharm Solutions Co Ltd
Bioasis Technologies Inc
Bioinnotis
Biom Therapeutics LLC
Biorchestra Co Ltd
Biovista Inc
Bloom Science Inc
Bright Minds Biosciences Inc
Bristol-Myers Squibb Co
CAMP4 Therapeutics Corp
Cann Pharmaceutical Australia Ltd
Cavion LLC
Celavie Biosciences LLC
Cellix Bio Pvt Ltd
Cerebral Therapeutics Inc
Cerecin Pte Ltd
Cerevel Therapeutics Holdings Inc
Coda Biotherapeutics Inc
Corestem Inc
Corlieve Therapeutics SAS
Crossject SA
CSL Ltd
Cure Pharmaceutical Holding Corp
CuroNZ Ltd
CURx Pharmaceuticals Inc
Cypralis Ltd
Eisai Co Ltd
Eliem Therapeutics Inc
Encoded Therapeutics Inc
Engrail Therapeutics Inc
Epalex Corp
EpyGenix Therapeutics Inc
Equilibre Biopharmaceuticals Corp
ES Therapeutics LLC
Eton Pharmaceuticals Inc
Ferring Pharmaceuticals Inc
GABA Therapeutics Inc
GAOMA Therapeutics
Gene Therapy Research Institution Co Ltd
GenEp Inc
Genochem SAS
Genus Lifesciences Inc
Guangzhou Cellprotek Pharmaceutical Co Ltd
H. Lundbeck AS
Harbin Pharmaceutical Group Co Ltd
Herophilus
ImmunoChem Therapeutics LLC
InteRNA Technologies BV
Ionis Pharmaceuticals Inc
Iproteos SL
iQure Pharma Inc
Jazz Pharmaceuticals Plc
JC Pharma Inc
Johnson & Johnson
Ketogen Inc
Klaria Pharma Holding AB
Knopp Biosciences LLC
LATITUDE Pharmaceuticals Inc
Lead Discovery Center GmbH
Lexaria Bioscience Corp
LifeSplice Pharma LLC
Lipocine Inc
Longboard Pharmaceuticals Inc
Luminus Biosciences Inc
Marinus Pharmaceuticals Inc
Medicure Inc
Mediphage Bioceuticals Inc
Medlab Clinical Ltd
Mercaptor Discoveries Inc
Metys Pharmaceuticals AG
MGC Pharmaceuticals Ltd
Mucodel Pharma LLC
MyMD Pharmaceuticals, Inc
MyX Therapeutics Inc
Nasus Pharma Ltd
Navega Therapeutics Inc
Navitor Pharmaceuticals Inc
Neonc Technologies Inc
NeuCyte Inc
NeuExcell Therapeutics Inc
NeurAegis Inc
Neuraxpharm France SAS
Neurelis Inc
NeuroAdjuvants Inc
Neurocrine Biosciences Inc
Neuroene Therapeutics LLC
Neurona Therapeutics Inc
New Amsterdam Sciences Inc
Nissan Chemical Corp
Noema Pharma AG
NoNO Inc
NuBioPharma LLC
OB Pharmaceuticals
OMass Therapeutics Limited
Oncocross Co Ltd
Otsuka Pharmaceutical Co Ltd
Ovid Therapeutics Inc
OWP Pharmaceuticals Inc
Panaxia Pharmaceutical Industries Ltd
Pfizer Inc
PharmaTher Holdings Ltd
PharmatrophiX Inc
PIQUR Therapeutics AG
Praxis Precision Medicines Inc
Primetime Life Sciences LLC
Promius Pharma LLC
Proniras Corp
Protheragen Inc
Proveca Ltd
PTC Therapeutics Inc
Puretech Health Plc
Q-State Biosciences Inc
Reata Pharmaceuticals Inc
reMYND NV
RespireRx Pharmaceuticals Inc
RogCon Inc
Sage Therapeutics Inc
Saniona AB
Scientus Pharma Inc
SciSparc Ltd
Serina Therapeutics Inc
Shackelford Pharma Inc
Shanghai Lvdao Pharmaceutical Technology Co Ltd
Shanghai Zhimeng Biopharma Inc
Simcere Pharmaceutical Group Ltd
SK Biopharmaceuticals Co Ltd
SK Chemicals Co Ltd
Skye Bioscience Inc
SoVarGen Co Ltd
Spark Therapeutics Inc
Stoke Therapeutics Inc
StrideBio Inc
Sumitomo Dainippon Pharma Co Ltd
Sun Pharma Advanced Research Company Ltd
Supernus Pharmaceuticals Inc
Sustained Nano Systems LLC
Taho Pharmaceuticals Ltd
Takeda Pharmaceutical Co Ltd
Tavanta Therapeutics Inc
Taysha Gene Therapies Inc
Techfields Pharma Co Ltd
Tevard Biosciences LLC
The Cell Factory BVBA
Trevena Inc
UCB SA
Ultragenyx Pharmaceutical Inc
Viatris Inc
Virpax Pharmaceuticals Inc
VistaGen Therapeutics Inc
Vitality Biopharma Inc
Vyripharm Biopharmaceuticals
Whan In Pharm Co Ltd
Xenon Pharmaceuticals Inc
Xeris Biopharma Holdings Inc
XPhyto Therapeutics Corp
XWPharma Ltd
Zhejiang Conba Pharmaceutical Co Ltd
Zogenix Inc
Zynerba Pharmaceuticals Inc
Table of Contents
Table
Figures
Frequently asked questions
-
What are the targets of the Epilepsy pipeline drugs market?
The targets of the epilepsy pipeline drugs market are Gamma-Aminobutyric Acid Type A Receptor Subunit (GABA, Voltage Gated Sodium Channel, Cannabinoid Receptor 2, Cannabinoid Receptor 1, Potassium Voltage Gated Channel Subfamily KQT Member 2, Potassium Voltage Gated Channel Subfamily KQT Member 3, Sodium Channel Protein Type 8 Subunit Alpha, Sodium Channel Protein Type 1 Subunit Alpha, 5-Hydroxytryptamine Receptor, and Serine/Threonine Protein Kinase mTOR.
-
What are the mechanisms of action of the Epilepsy pipeline drugs market?
The mechanisms of action of the epilepsy pipeline drugs market are Gamma-Aminobutyric Acid Type A Receptor Subunit (GABAAgonist, Voltage Gated Sodium Channel Blocker, Cannabinoid Receptor 2 Agonist, Cannabinoid Receptor 1 Agonist, Potassium Voltage Gated Channel Subfamily KQT Member 2 Activator, Potassium Voltage Gated Channel Subfamily KQT Member 3 Activator, Sodium Channel Protein Type 1 Subunit Alpha Activator, Sodium Channel Protein Type 8 Subunit Alpha Blocker, 5-Hydroxytryptamine Receptor Agonist, and Serine/Threonine Protein Kinase mTOR Inhibitor.
-
What are the routes of administration in the Epilepsy pipeline drugs market?
The routes of administration in the epilepsy pipeline drugs market are oral, intravenous, subcutaneous, nasal, intrathecal, buccal, inhalational, sublingual, transdermal, and parenteral.
-
What are the molecule types in the Epilepsy pipeline drugs market?
The molecule types in the epilepsy pipeline drugs market are small molecule, antisense oligonucleotide, gene therapy, synthetic peptide, cell therapy, RNAi gene therapy, antibody, antisense RNAi oligonucleotide, biologic, and fusion protein.
-
Which are the key companies in the Epilepsy pipeline drugs market?
Some of the key companies in the epilepsy pipeline drugs market are Praxis Precision Medicines Inc, EpyGenix Therapeutics Inc, Neurocrine Biosciences Inc, Bio-Pharm Solutions Co Ltd, Q-State Biosciences Inc, Taysha Gene Therapies Inc, UCB SA, Jazz Pharmaceuticals Plc, SK Biopharmaceuticals Co Ltd, and AfaSci Inc.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more Central Nervous System reports