Glucose Monitoring – Pipeline Products by Stage of Development 34
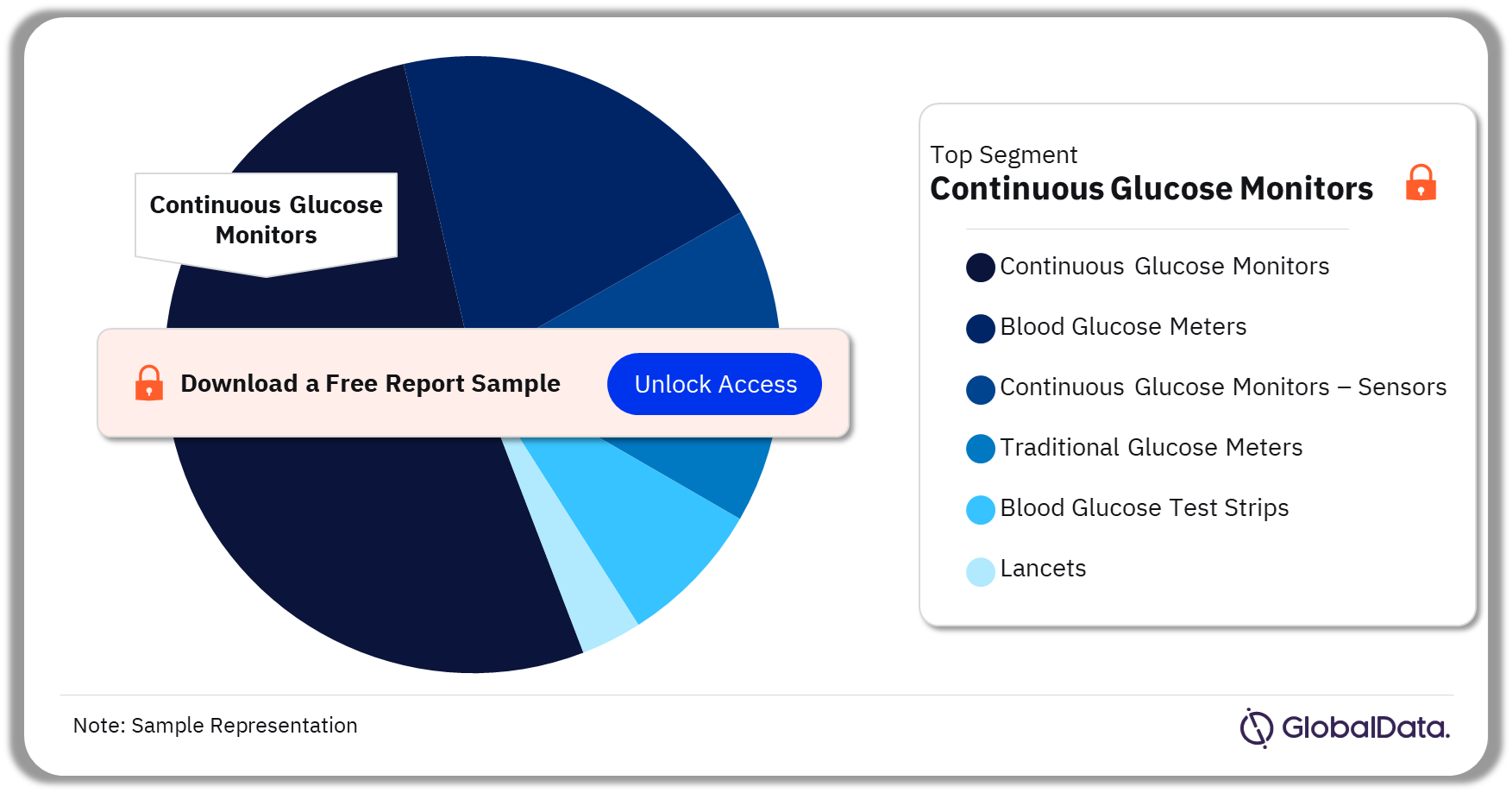
Glucose Monitoring – Pipeline Products by Segment 35
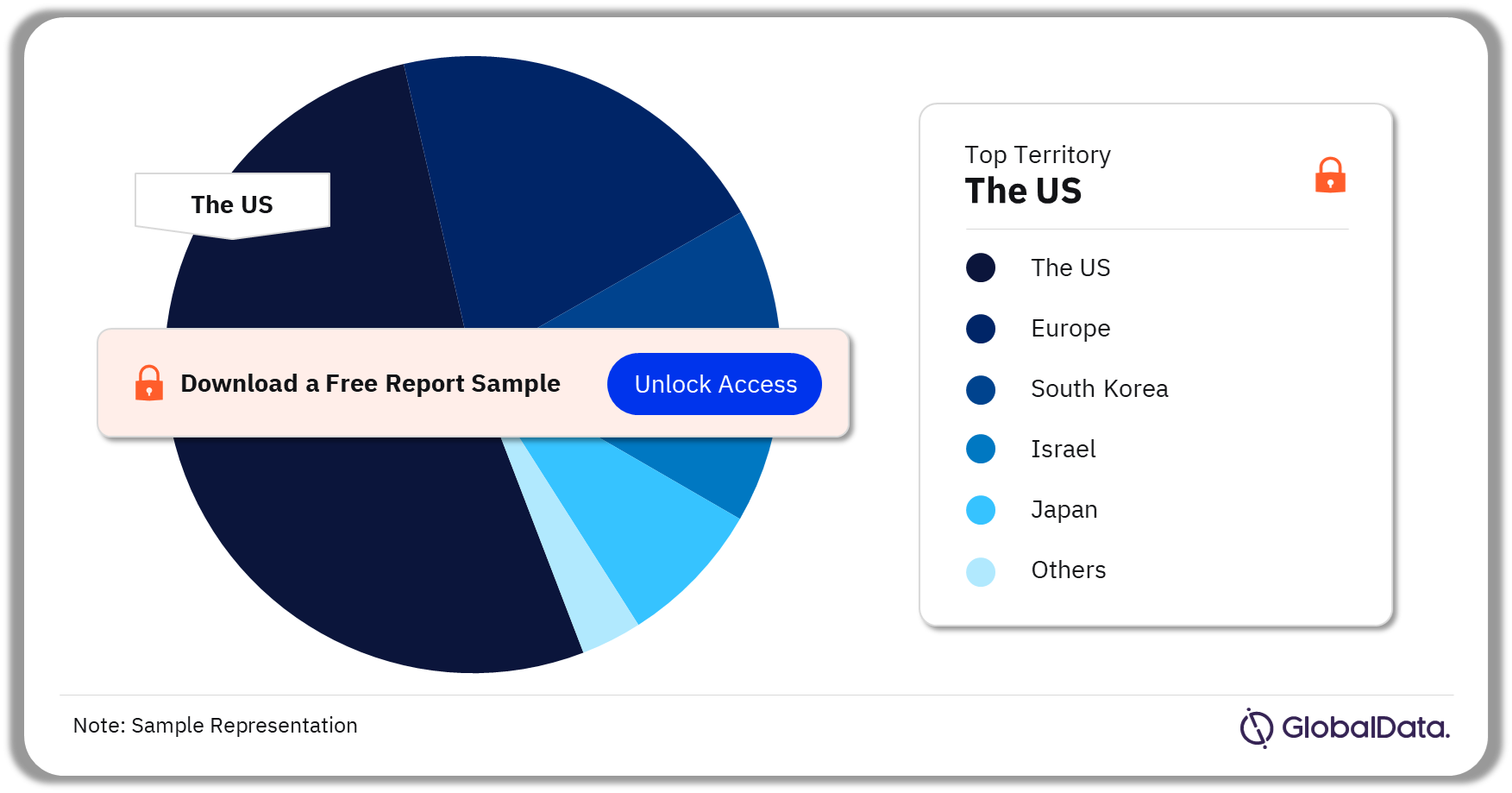
Glucose Monitoring – Pipeline Products by Territory 36
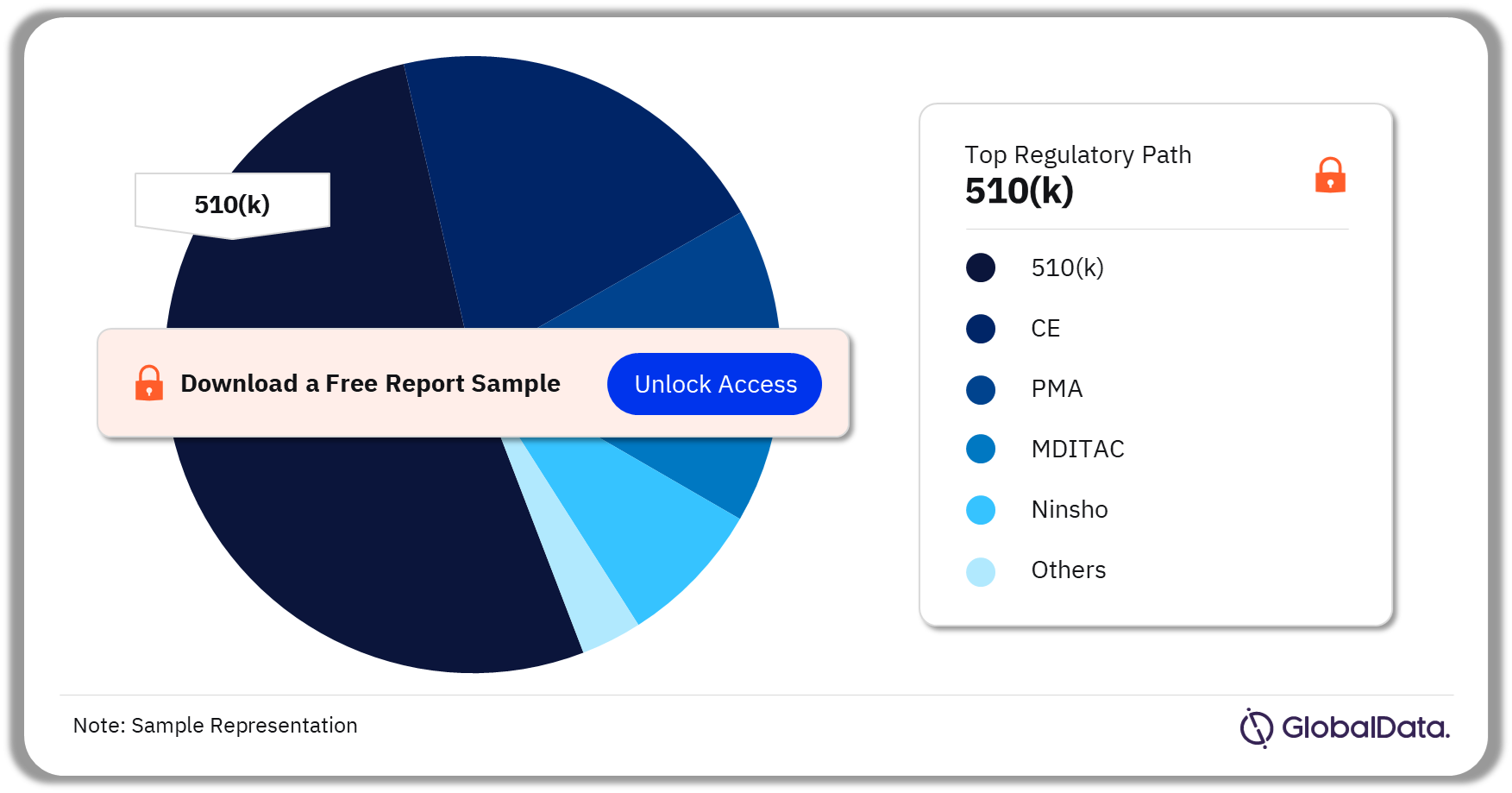
Glucose Monitoring – Pipeline Products by Regulatory Path 38
Glucose Monitoring – Pipeline Products by Estimated Approval Date 39
Glucose Monitoring – Ongoing Clinical Trials 40
Glucose Monitoring Companies – Pipeline Products by Stage of Development 41
Glucose Monitoring – Pipeline Products by Stage of Development 50
4a Medicom GmbH Pipeline Products & Ongoing Clinical Trials Overview 58
GlucoPEARL – Product Status 58
GlucoPEARL – Product Description 58
Abbott Diabetes Care Inc Pipeline Products & Ongoing Clinical Trials Overview 59
FreeStyle Libre Flash Glucose Monitoring System – Children – Product Status 59
FreeStyle Libre Flash Glucose Monitoring System – Children – Product Description 59
Fully Integrated Blood Glucose Monitoring System – Product Status 60
Fully Integrated Blood Glucose Monitoring System – Product Description 60
Modified FreeStyle Libre 2 Flash Glucose Monitoring Sensor – Pregnant Women – Product Status 60
Modified FreeStyle Libre 2 Flash Glucose Monitoring Sensor – Pregnant Women – Product Description 61
Modified FreeStyle Libre 3 Flash Glucose Monitoring Sensor – Pregnant Women – Product Status 61
Modified FreeStyle Libre 3 Flash Glucose Monitoring Sensor – Pregnant Women – Product Description 61
Updated Hospital Blood Glucose Monitoring System – Product Status 62
Updated Hospital Blood Glucose Monitoring System – Product Description 62
Abbott Healthcare Pvt Ltd Pipeline Products & Ongoing Clinical Trials Overview 63
Lingo Glucose – Product Status 63
Lingo Glucose – Product Description 63
Abbott Laboratories Pipeline Products & Ongoing Clinical Trials Overview 64
Continuous Glucose-Ketone Monitoring System – Product Status 64
Continuous Glucose-Ketone Monitoring System – Product Description 65
Accessible Diagnostics LLC Pipeline Products & Ongoing Clinical Trials Overview 66
Life-Tab – Product Status 66
Life-Tab – Product Description 66
Achira Labs Pvt Ltd Pipeline Products & Ongoing Clinical Trials Overview 67
Silk Strip – Diabetes – Product Status 67
Silk Strip – Diabetes – Product Description 67
Actxa Pte Ltd Pipeline Products & Ongoing Clinical Trials Overview 68
Actxa BGM Tracker (GLO2) – Product Status 68
Actxa BGM Tracker (GLO2) – Product Description 68
SVT In-Ear – Product Status 69
SVT In-Ear – Product Description 69
Advanced Biosensors-Ohio LLC Pipeline Products & Ongoing Clinical Trials Overview 70
My Medibit – Product Status 70
My Medibit – Product Description 70
Advanced Medical Electronics Corp Pipeline Products & Ongoing Clinical Trials Overview 71
Continuous Glucose Monitor – Product Status 71
Continuous Glucose Monitor – Product Description 71
Afon Technology Ltd Pipeline Products & Ongoing Clinical Trials Overview 72
Non-Invasive Blood Glucose Monitor – Product Status 72
Non-Invasive Blood Glucose Monitor – Product Description 72
Afon Technology Ltd – Ongoing Clinical Trials Overview 73
Non-Invasive Blood Glucose Monitor – Development Study to Undertake a Comprehensive Performance Evaluation of Prototype Non-Invasive Blood Glucose Measurement Systems Compared to a Standard (Invasive) Measurement Method of Blood Glucose 74
Alertgy Inc Pipeline Products & Ongoing Clinical Trials Overview 75
Alertgy NICGM – Product Status 75
Alertgy NICGM – Product Description 75
All India Institute of Medical Sciences Pipeline Products & Ongoing Clinical Trials Overview 76
Standalone Meter – Glucose Quantification – Product Status 76
Standalone Meter – Glucose Quantification – Product Description 76
ALR Technologies Inc Pipeline Products & Ongoing Clinical Trials Overview 77
Bluetooth Enabled Complementary Blood Glucose Meter – Product Status 77
Bluetooth Enabled Complementary Blood Glucose Meter – Product Description 77
Complementary Blood Glucose Test Strip – Product Status 78
Complementary Blood Glucose Test Strip – Product Description 78
American Chemical Society Pipeline Products & Ongoing Clinical Trials Overview 79
Touch-Based Sweat Glucose Sensor – Product Status 79
Touch-Based Sweat Glucose Sensor – Product Description 79
American Chemical Society – Ongoing Clinical Trials Overview 80
Touch-Based Sweat Glucose Sensor – Personalized Fingertip Glucose Measurement With a Touch Sensor 81
Arizona State University Pipeline Products & Ongoing Clinical Trials Overview 82
TOUCH Tears Device – Product Status 82
TOUCH Tears Device – Product Description 82
Arkal Medical Inc (Inactive) Pipeline Products & Ongoing Clinical Trials Overview 83
GlucoPod CGM System – Product Status 83
GlucoPod CGM System – Product Description 83
Azurite Technologies, Inc. (Inactive) Pipeline Products & Ongoing Clinical Trials Overview 84
Blood Glucose Sensor – Product Status 84
Blood Glucose Sensor – Product Description 84
Bar-Ilan University Pipeline Products & Ongoing Clinical Trials Overview 85
Biometric Watch – Product Status 85
Biometric Watch – Product Description 85
Baylor University Pipeline Products & Ongoing Clinical Trials Overview 86
Portable Non-Invasive Blood Glucose Monitor – Product Status 86
Portable Non-Invasive Blood Glucose Monitor – Product Description 86
Biofi Medical Healthcare Pvt Ltd Pipeline Products & Ongoing Clinical Trials Overview 87
Non-Invasive Blood Glucometer – Product Status 87
Non-Invasive Blood Glucometer – Product Description 87
BioGliko Sp zoo Pipeline Products & Ongoing Clinical Trials Overview 88
Glucomfort – Product Status 88
Glucomfort – Product Description 88
Biolinq Inc Pipeline Products & Ongoing Clinical Trials Overview 89
Intelligent Continuous Glucose Monitoring System – Product Status 89
Intelligent Continuous Glucose Monitoring System – Product Description 89
Biopeak Corp Pipeline Products & Ongoing Clinical Trials Overview 90
Non-Invasive Glucose Analyzer – Product Status 90
Non-Invasive Glucose Analyzer – Product Description 90
Biorasis Inc Pipeline Products & Ongoing Clinical Trials Overview 91
Glucowizzard – Product Status 91
Glucowizzard – Product Description 91
Biosense Technologies Pipeline Products & Ongoing Clinical Trials Overview 92
B-myWatch – Product Status 92
B-myWatch – Product Description 92
BioTex Inc Pipeline Products & Ongoing Clinical Trials Overview 93
Smart Tattoo Glucose Sensor – Product Status 93
Smart Tattoo Glucose Sensor – Product Description 93
Biovotion AG Pipeline Products & Ongoing Clinical Trials Overview 94
VSM 3 Glucose Monitoring System – Product Status 94
VSM 3 Glucose Monitoring System – Product Description 94
BlueVine Graphene Industries, Inc. Pipeline Products & Ongoing Clinical Trials Overview 95
First-Generation Glucose Monitoring Device – Product Status 95
First-Generation Glucose Monitoring Device – Product Description 95
Noninvasive Glucose Monitoring Device – Product Status 96
Noninvasive Glucose Monitoring Device – Product Description 96
Brain Beat Ltd Pipeline Products & Ongoing Clinical Trials Overview 97
Edvais – Product Status 97
Edvais – Product Description 97
Brigham and Women’s Hospital Pipeline Products & Ongoing Clinical Trials Overview 98
Continuous Glucose Monitoring Sensor – Product Status 98
Continuous Glucose Monitoring Sensor – Product Description 98
Brighter AB Pipeline Products & Ongoing Clinical Trials Overview 99
Actiste – Product Status 99
Actiste – Product Description 99
Actiste Mini – Product Status 100
Actiste Mini – Product Description 100
Brighter AB – Ongoing Clinical Trials Overview 101
Actiste – A Pilot User Experience Study to Assess Actiste Diabetes Management as a Service compared to Patient’s Conventional Diabetes Management 102
CALISTO MEDICAL, INC. (Inactive) Pipeline Products & Ongoing Clinical Trials Overview 103
Glucoband – Product Status 103
Glucoband – Product Description 103
Callitas Therapeutics Inc Pipeline Products & Ongoing Clinical Trials Overview 104
Electronic Mosquito – Product Status 104
Electronic Mosquito – Product Description 104
Cardiff University Pipeline Products & Ongoing Clinical Trials Overview 105
Non-Invasive Blood Glucose Meter – Product Status 105
Non-Invasive Blood Glucose Meter – Product Description 105
Cascade Metrix LLC Pipeline Products & Ongoing Clinical Trials Overview 106
AutoPlexer – Product Status 106
AutoPlexer – Product Description 106
CGM3 Ltd Pipeline Products & Ongoing Clinical Trials Overview 107
Miniaturized Implantable Continuous Glucose Monitor – Product Status 107
Miniaturized Implantable Continuous Glucose Monitor – Product Description 107
Cirtec Medical Corp Pipeline Products & Ongoing Clinical Trials Overview 108
Blood Glucose Monitor – Product Status 108
Blood Glucose Monitor – Product Description 108
City, University of London Pipeline Products & Ongoing Clinical Trials Overview 109
Non-Invasive Glucose Monitor – Product Status 109
Non-Invasive Glucose Monitor – Product Description 109
Columbia University Pipeline Products & Ongoing Clinical Trials Overview 110
Wearable Continuous Blood Glucose Monitoring Device – Product Status 110
Wearable Continuous Blood Glucose Monitoring Device – Product Description 110
ConvaTec Group Plc Pipeline Products & Ongoing Clinical Trials Overview 111
Closed Loop Continuous Glucose Monitoring System – Product Status 111
Closed Loop Continuous Glucose Monitoring System – Product Description 111
Co-Optical Inc Pipeline Products & Ongoing Clinical Trials Overview 112
Co-Optical Device – Product Status 112
Co-Optical Device – Product Description 112
Cyber Medical Ltd (Inactive) Pipeline Products & Ongoing Clinical Trials Overview 113
Cybersensors Continuous Glucose Sensor – Product Status 113
Cybersensors Continuous Glucose Sensor – Product Description 113
DexCom Inc Pipeline Products & Ongoing Clinical Trials Overview 114
Dexcom 15 Day Wear Sensor – Product Status 114
Dexcom 15 Day Wear Sensor – Product Description 115
DexCom CGM With Accu-Chek Insulin Delivery System – Product Status 115
DexCom CGM With Accu-Chek Insulin Delivery System – Product Description 115
Dexcom G7 CGM System – Product Status 116
Dexcom G7 CGM System – Product Description 117
Dexcom Hospital Continuous Glucose Monitoring System – Product Status 117
Dexcom Hospital Continuous Glucose Monitoring System – Product Description 118
First-Gen Verily Sensor – Product Status 118
First-Gen Verily Sensor – Product Description 118
DexCom Inc – Ongoing Clinical Trials Overview 119
DexCom CGM With Accu-Chek Insulin Delivery System – Evaluation of a Closed-loop System for the Treatment of Type 2 Diabetes at Home (DT2_2) 120
DiaMonTech GmbH Pipeline Products & Ongoing Clinical Trials Overview 121
D-Band – Product Status 121
D-Band – Product Description 121
D-Pocket – Product Status 122
D-Pocket – Product Description 122
DIRAmed LLC. Pipeline Products & Ongoing Clinical Trials Overview 123
Glucose Monitor – Product Status 123
Glucose Monitor – Product Description 123
Glucose Monitor – Home Use – Product Status 124
Glucose Monitor – Home Use – Product Description 124
DirectSens GmbH Pipeline Products & Ongoing Clinical Trials Overview 125
Blood Glucose Test Strip – Product Status 125
Blood Glucose Test Strip – Product Description 125
Glucose Sensor – Product Status 126
Glucose Sensor – Product Description 126
Dongwoon Anatech Co Ltd Pipeline Products & Ongoing Clinical Trials Overview 127
D-SaLife – Product Status 127
D-SaLife – Product Description 127
Drawbridge Health Inc Pipeline Products & Ongoing Clinical Trials Overview 128
OneDraw A1C Test System – Product Status 128
OneDraw A1C Test System – Product Description 128
Drawbridge Health Inc – Ongoing Clinical Trials Overview 129
OneDraw A1C Test System – Study to Evaluate the Usage of OneDraw Blood Collection Device for Remote Blood Sample Collection 130
Duquesne University Pipeline Products & Ongoing Clinical Trials Overview 131
Electrochemical Glucose Sensor – Product Status 131
Electrochemical Glucose Sensor – Product Description 131
Eccrine Systems Inc Pipeline Products & Ongoing Clinical Trials Overview 132
Sweatronics Electronic Patch – Product Status 132
Sweatronics Electronic Patch – Product Description 132
Echo Therapeutics Inc Pipeline Products & Ongoing Clinical Trials Overview 133
Display – Continuous Glucose Monitoring – Product Status 133
Display – Continuous Glucose Monitoring – Product Description 133
Gen 2 Symphony CGM System – Product Status 134
Gen 2 Symphony CGM System – Product Description 134
SENSOR – New Flexible Glucose Sensor & Transmitter – Product Status 134
SENSOR – New Flexible Glucose Sensor & Transmitter – Product Description 135
Symphony tCGM System – Home – Product Status 135
Symphony tCGM System – Home – Product Description 135
Symphony tCGM System – Hospital Critical Care – Product Status 136
Symphony tCGM System – Hospital Critical Care – Product Description 136
Ecole Polytechnique Federale de Lausanne Pipeline Products & Ongoing Clinical Trials Overview 137
Viscosimetric Glucose Sensor – Product Status 137
Viscosimetric Glucose Sensor – Product Description 137
eLutions Integrated Systems, Inc. Pipeline Products & Ongoing Clinical Trials Overview 138
Optically-Efficient Miniaturized Raman Optical System – Product Status 138
Optically-Efficient Miniaturized Raman Optical System – Product Description 138
Emperra GmbH E-Health Technologies Pipeline Products & Ongoing Clinical Trials Overview 139
GSM Enabled Blood Glucose Meter – Product Status 139
GSM Enabled Blood Glucose Meter – Product Description 139
Epinex Diagnostics Inc Pipeline Products & Ongoing Clinical Trials Overview 140
Epinex G1A OTC Device – Product Status 140
Epinex G1A OTC Device – Product Description 140
Epinex G1A POC Device – Product Status 141
Epinex G1A POC Device – Product Description 141
Eternity Healthcare Inc Pipeline Products & Ongoing Clinical Trials Overview 142
Dual Saliva-Based Sugar Monitor – Product Status 142
Dual Saliva-Based Sugar Monitor – Product Description 142
EyeSense GmbH Pipeline Products & Ongoing Clinical Trials Overview 143
Eye Sensor – Product Status 143
Eye Sensor – Product Description 143
FiberSense System – Product Status 144
FiberSense System – Product Description 144
EyeSense GmbH – Ongoing Clinical Trials Overview 145
FiberSense System – Effectiveness and Safety Study of the Continuous Glucose Monitoring System for Home Use (Including In-clinic Sessions) in Patients with Diabetes 146
FiberSense System – Effectiveness and Safety Study of the Percutaneous Optical Fibre Glucose Sensor (FiberSense) for Home Use in Diabetic and Prediabetic Individuals 146
FiberSense System – Effectiveness and Safety Study to Investigate the Improved FiberSense Continuous Glucose Monitoring System in Diabetic Patients 146
Firefly Health Pty Ltd Pipeline Products & Ongoing Clinical Trials Overview 147
FireFly – Product Status 147
FireFly – Product Description 147
Flexible Medical Systems, LLC Pipeline Products & Ongoing Clinical Trials Overview 148
Biomonitoring System – Product Status 148
Biomonitoring System – Product Description 148
Flowsion A/S Pipeline Products & Ongoing Clinical Trials Overview 149
Glycostat – Product Status 149
Glycostat – Product Description 149
Fluorometrix Corp Pipeline Products & Ongoing Clinical Trials Overview 150
Neonatal Glucose Monitor – Product Status 150
Neonatal Glucose Monitor – Product Description 150
Gentag Inc Pipeline Products & Ongoing Clinical Trials Overview 151
Pain-Free Diabetes Monitoring Patch – Product Status 151
Pain-Free Diabetes Monitoring Patch – Product Description 151
Georgia Institute of Technology Pipeline Products & Ongoing Clinical Trials Overview 152
Glucose Regulation Device – Product Status 152
Glucose Regulation Device – Product Description 152
Gili Medical Ltd Pipeline Products & Ongoing Clinical Trials Overview 153
Diawatch – Product Status 153
Diawatch – Product Description 153
Globe Medical Tech Inc (Inactive) Pipeline Products & Ongoing Clinical Trials Overview 154
Continuous Glucose Monitoring Device – Product Status 154
Continuous Glucose Monitoring Device – Product Description 154
Gluco-Chaser Inc Pipeline Products & Ongoing Clinical Trials Overview 155
Disposable Strip BGM Prototype Sensor – Product Status 155
Disposable Strip BGM Prototype Sensor – Product Description 155
GlucoJet Pipeline Products & Ongoing Clinical Trials Overview 156
Glucose Sensor – Product Status 156
Glucose Sensor – Product Description 156
GlucoLight Corporation (Inactive) Pipeline Products & Ongoing Clinical Trials Overview 157
SENTRIS-100 – Product Status 157
SENTRIS-100 – Product Description 157
GlucoMe Ltd Pipeline Products & Ongoing Clinical Trials Overview 158
GlucoMe Glucose Monitoring Device – Product Status 158
GlucoMe Glucose Monitoring Device – Product Description 158
Glucometrix AG Pipeline Products & Ongoing Clinical Trials Overview 159
Non-Invasive Blood Glucose Monitoring Device – Product Status 159
Non-Invasive Blood Glucose Monitoring Device – Product Description 159
GlucoModicum Oy Pipeline Products & Ongoing Clinical Trials Overview 160
Talisman – Product Status 160
Talisman – Product Description 160
Glucorecs Pipeline Products & Ongoing Clinical Trials Overview 161
GluFit – Product Status 161
GluFit – Product Description 161
GlucoRx Ltd Pipeline Products & Ongoing Clinical Trials Overview 162
GlucoRx BioXensor – Product Status 162
GlucoRx BioXensor – Product Description 162
GlucosAlarm Pipeline Products & Ongoing Clinical Trials Overview 163
GlucosAlarm – Product Status 163
GlucosAlarm – Product Description 163
Glucosat Pipeline Products & Ongoing Clinical Trials Overview 164
Glucosat – Product Status 164
Glucosat – Product Description 164
Glucosense Diagnostics Ltd (Inactive) Pipeline Products & Ongoing Clinical Trials Overview 165
Non-Invasive Blood Glucose Monitor – Product Status 165
Non-Invasive Blood Glucose Monitor – Product Description 165
Non-Invasive Blood Glucose Monitor – Finger-touch Version – Product Status 166
Non-Invasive Blood Glucose Monitor – Finger-touch Version – Product Description 166
GlucoSet AS Pipeline Products & Ongoing Clinical Trials Overview 167
GlucoSet Sensor – Product Status 167
GlucoSet Sensor – Product Description 167
GlucoStats System Pte Ltd. Pipeline Products & Ongoing Clinical Trials Overview 168
First Generation Glucose Monitoring Device – Product Status 168
First Generation Glucose Monitoring Device – Product Description 168
Second Generation Glucose Monitoring Device – Product Status 169
Second Generation Glucose Monitoring Device – Product Description 169
GlucoTrack Inc Pipeline Products & Ongoing Clinical Trials Overview 170
GlucoTrack – DF-C – Product Status 170
GlucoTrack – DF-C – Product Description 171
GlucoTrack – DF-D – Product Status 171
GlucoTrack – DF-D – Product Description 171
GlucoTrack – DF-I – Product Status 172
GlucoTrack – DF-I – Product Description 172
GlucoTrack – DF-N – Product Status 172
GlucoTrack – DF-N – Product Description 173
GlucoTrack – DF-P – Product Status 173
GlucoTrack – DF-P – Product Description 173
GlucoTrack DF-F – Product Status 174
GlucoTrack DF-F – Product Description 174
Second Generation Glucotrack – Product Status 174
Second Generation Glucotrack – Product Description 175
Glucovation Inc Pipeline Products & Ongoing Clinical Trials Overview 176
GW Wearable Glucose Sensor – Product Status 176
GW Wearable Glucose Sensor – Product Description 176
GluMetrics Inc Pipeline Products & Ongoing Clinical Trials Overview 177
GluCath Intravascular Continuous Glucose Monitor – Product Status 177
GluCath Intravascular Continuous Glucose Monitor – Product Description 177
Glusense Ltd Pipeline Products & Ongoing Clinical Trials Overview 178
Glyde CGM – Product Status 178
Glyde CGM – Product Description 178
Glusense Ltd – Ongoing Clinical Trials Overview 179
Glyde CGM – A Study to Evaluate the Safety and Efficacy of the GluSense’s Glyde CGM Technology in Humans 180
GlySens Inc Pipeline Products & Ongoing Clinical Trials Overview 181
Eclipse 3 ICGM System – Product Status 181
Eclipse 3 ICGM System – Product Description 181
Eclipse ICGM System – Product Status 182
Eclipse ICGM System – Product Description 182
Second Generation ICGM System – Product Status 182
Second Generation ICGM System – Product Description 183
GlySens Inc – Ongoing Clinical Trials Overview 184
Eclipse 3 ICGM System – Feasibility Trial to Evaluate the Eclipse 3 Continuous Glucose Monitoring System for Individuals with Type 1 Diabetes Mellitus – Australia (FREE – AU Trial) 185
GOQii Technologies Pvt Ltd Pipeline Products & Ongoing Clinical Trials Overview 186
GOQii SugarWise – Product Status 186
GOQii SugarWise – Product Description 186
GOQii Technologies Pvt Ltd – Ongoing Clinical Trials Overview 187
GOQii SugarWise – A Study to Validate the GOQii SugarWise Non-invasive Innovative Diabetic Risk Monitoring System 188
G-Sense Ltd. Pipeline Products & Ongoing Clinical Trials Overview 189
G-Sense Device – Product Status 189
G-Sense Device – Product Description 189
Guangzhou Powder Pharmaceuticals Ltd Pipeline Products & Ongoing Clinical Trials Overview 190
Continuous Glucose Monitoring System – Product Status 190
Continuous Glucose Monitoring System – Product Description 190
Guided Therapeutics Inc Pipeline Products & Ongoing Clinical Trials Overview 191
Altea MicroPor Laser – Product Status 191
Altea MicroPor Laser – Product Description 191
Hagar Tech Pipeline Products & Ongoing Clinical Trials Overview 192
GWave – Product Status 192
GWave – Product Description 192
GWave 2.0 – Product Status 193
GWave 2.0 – Product Description 193
Hagar Tech – Ongoing Clinical Trials Overview 194
GWave – Study to Evaluate the Accuracy of GWave in Patients with Type 1 and Type 2 diabetes in Israel 195
Hanyang University Pipeline Products & Ongoing Clinical Trials Overview 196
Colorimetric Contact Lens Sensor – Product Status 196
Colorimetric Contact Lens Sensor – Product Description 196
Helo Corp Pipeline Products & Ongoing Clinical Trials Overview 197
Helo LX Pro – Product Status 197
Helo LX Pro – Product Description 197
iGlyko, Inc. (Inactive) Pipeline Products & Ongoing Clinical Trials Overview 198
iGlyko System – Product Status 198
iGlyko System – Product Description 198
Indian Institute of Technology Delhi Pipeline Products & Ongoing Clinical Trials Overview 199
Non-Invasive Glucose Monitoring System – Product Status 199
Non-Invasive Glucose Monitoring System – Product Description 199
Indiana University Pipeline Products & Ongoing Clinical Trials Overview 200
Breathalyzer-Type Low Blood Sugar Warning Device – Product Status 200
Breathalyzer-Type Low Blood Sugar Warning Device – Product Description 200
Indigo Diabetes NV Pipeline Products & Ongoing Clinical Trials Overview 201
Continuous Multi-Metabolite Monitoring Sensor – Product Status 201
Continuous Multi-Metabolite Monitoring Sensor – Product Description 201
Informed Data Systems Inc Pipeline Products & Ongoing Clinical Trials Overview 202
Disposable Biowearable Sensor – Product Status 202
Disposable Biowearable Sensor – Product Description 202
InLight Solutions Inc. Pipeline Products & Ongoing Clinical Trials Overview 203
Non-Invasive Glucose Measurement System – Product Status 203
Non-Invasive Glucose Measurement System – Product Description 203
Innopharmax Inc Pipeline Products & Ongoing Clinical Trials Overview 204
P12001 – Product Status 204
P12001 – Product Description 204
Innova Medical Design Pipeline Products & Ongoing Clinical Trials Overview 205
Diabetes Lancet and Injection Pain Relief Device – Product Status 205
Diabetes Lancet and Injection Pain Relief Device – Product Description 205
Innovosens AB Pipeline Products & Ongoing Clinical Trials Overview 206
Diabetic Sensor – Continuous Glucose Monitoring – Product Status 206
Diabetic Sensor – Continuous Glucose Monitoring – Product Description 206
Integrated Medical Sensors Pipeline Products & Ongoing Clinical Trials Overview 207
Implantable Continuous Glucose Monitor – Product Status 207
Implantable Continuous Glucose Monitor – Product Description 207
IntelliDx Inc (Inactive) Pipeline Products & Ongoing Clinical Trials Overview 208
Optimus – Product Status 208
Optimus – Product Description 208
Intelligent Bio Solutions Inc Pipeline Products & Ongoing Clinical Trials Overview 209
Saliva Glucose Biosensor – Product Status 210
Saliva Glucose Biosensor – Product Description 211
Intelligent Bio Solutions Inc – Ongoing Clinical Trials Overview 212
Saliva Glucose Biosensor – Specimen Collection Methodology to Support Subsequent Correlation Analyses of Glucose in Oral Fluids, Capillary and Venous Blood 213
InventHelp Pipeline Products & Ongoing Clinical Trials Overview 214
Meter Reader – Product Status 214
Meter Reader – Product Description 214
Invivomon Inc (Inactive) Pipeline Products & Ongoing Clinical Trials Overview 215
Smart IV – Glucose Monitoring System – Product Status 215
Smart IV – Glucose Monitoring System – Product Description 215
Invivosense ASA (Inactive) Pipeline Products & Ongoing Clinical Trials Overview 216
IvSCGM Biosensor – Product Status 216
IvSCGM Biosensor – Product Description 216
I-Sens Inc Pipeline Products & Ongoing Clinical Trials Overview 217
CareSens Air Glucose Monitoring System – Product Status 217
CareSens Air Glucose Monitoring System – Product Description 217
Continuous Blood Glucose Meter – Product Status 218
Continuous Blood Glucose Meter – Product Description 218
iSense Corporation (Inactive) Pipeline Products & Ongoing Clinical Trials Overview 219
iSense CGM – Product Status 219
iSense CGM – Product Description 219
Jiangsu Yuyue Medical Equipment & Supply Co Ltd Pipeline Products & Ongoing Clinical Trials Overview 220
Second-Generation CGM System – Product Status 220
Second-Generation CGM System – Product Description 220
KeenCAT Technologies Pipeline Products & Ongoing Clinical Trials Overview 221
GlucoCAT – Product Status 221
GlucoCAT – Product Description 221
Keratin Biosciences Inc Pipeline Products & Ongoing Clinical Trials Overview 222
Illume – Product Status 222
Illume – Product Description 222
Know Labs Inc Pipeline Products & Ongoing Clinical Trials Overview 223
KnowU – Product Status 223
KnowU – Product Description 223
UBAND – Product Status 224
UBAND – Product Description 224
KTH Royal Institute of Technology Pipeline Products & Ongoing Clinical Trials Overview 225
Microneedle Patch – Glucose Monitoring – Product Status 225
Microneedle Patch – Glucose Monitoring – Product Description 225
Kumetrix, Inc. Pipeline Products & Ongoing Clinical Trials Overview 226
Silicon Micro Needle – Product Status 226
Silicon Micro Needle – Product Description 226
Laxmi Therapeutic Devices Inc Pipeline Products & Ongoing Clinical Trials Overview 227
Microneedle-Based Continuous Glucose Monitor – Product Status 227
Microneedle-Based Continuous Glucose Monitor – Product Description 227
Lein Applied Diagnostics Limited Pipeline Products & Ongoing Clinical Trials Overview 228
One Look – Product Status 228
One Look – Product Description 228
LEMM Technologies, LLC Pipeline Products & Ongoing Clinical Trials Overview 229
Non-Invasive Blood Glucose Monitor – Product Status 229
Non-Invasive Blood Glucose Monitor – Product Description 229
Lifecare AS Pipeline Products & Ongoing Clinical Trials Overview 230
Sencell Osmotic Pressure Sensor – Product Status 230
Sencell Osmotic Pressure Sensor – Product Description 230
LifeScan Inc Pipeline Products & Ongoing Clinical Trials Overview 231
CGM Sensor – Product Status 231
CGM Sensor – Product Description 231
Next Generation OneTouch UltraVue Verio Platform Glucose Testing System – Product Status 232
Next Generation OneTouch UltraVue Verio Platform Glucose Testing System – Product Description 232
ONETOUCH VERIO Pro+ POC Glucose Testing System – Version 1.5 – Product Status 232
ONETOUCH VERIO Pro+ POC Glucose Testing System – Version 1.5 – Product Description 232
Ligalli BV Pipeline Products & Ongoing Clinical Trials Overview 233
LIG MR19 – Product Status 233
LIG MR19 – Product Description 233
Light Touch Medical Inc Pipeline Products & Ongoing Clinical Trials Overview 234
Ocular Tear Pen – Product Status 234
Ocular Tear Pen – Product Description 234
Light Touch Technology Co Ltd Pipeline Products & Ongoing Clinical Trials Overview 235
Carbo Health Monitor – Product Status 235
Carbo Health Monitor – Product Description 235
Mobile Blood Glucose Sensor – Product Status 236
Mobile Blood Glucose Sensor – Product Description 236
LighTouch Medical, Inc. Pipeline Products & Ongoing Clinical Trials Overview 237
LighTouch Glucose Monitor – Product Status 237
LighTouch Glucose Monitor – Product Description 237
LighTouch Medical, Inc. – Ongoing Clinical Trials Overview 238
LighTouch Glucose Monitor – In Vivo Raman Spectroscopy (IVRS) of Human Fingertip Capillary Beds 239
Luminous Medical Inc (Inactive) Pipeline Products & Ongoing Clinical Trials Overview 240
Argus Glucose Monitoring System – Product Status 240
Argus Glucose Monitoring System – Product Description 240
Lyte Labs Pipeline Products & Ongoing Clinical Trials Overview 241
Wearable Smart Ring – Product Status 241
Wearable Smart Ring – Product Description 241
Major Biosystem Corp Pipeline Products & Ongoing Clinical Trials Overview 242
MajorCare AP-5100 – Strip Ejection – Product Status 242
MajorCare AP-5100 – Strip Ejection – Product Description 242
Massachusetts Institute of Technology Pipeline Products & Ongoing Clinical Trials Overview 243
Implantable Glucose Sensor – Product Status 243
Implantable Glucose Sensor – Product Description 243
Non-Invasive Blood Glucose Monitor – Product Status 244
Non-Invasive Blood Glucose Monitor – Product Description 244
Medella Health Inc. Pipeline Products & Ongoing Clinical Trials Overview 245
Smart Contact Lens – Product Status 245
Smart Contact Lens – Product Description 245
Medical Wireless Sensing Ltd Pipeline Products & Ongoing Clinical Trials Overview 246
GlucoWiSe – Product Status 246
GlucoWiSe – Product Description 246
Medtronic MiniMed Inc Pipeline Products & Ongoing Clinical Trials Overview 247
Combo Set Glucose Monitoring System – Product Status 247
Combo Set Glucose Monitoring System – Product Description 247
Comfort Sensor – Product Status 248
Comfort Sensor – Product Description 248
Harmony 1 Sensor – Product Status 248
Harmony 1 Sensor – Product Description 248
Harmony 2 Sensor – Product Status 249
Harmony 2 Sensor – Product Description 249
iPro 3 Glucose Monitoring System – Product Status 249
iPro 3 Glucose Monitoring System – Product Description 250
ipro 4 Glucose Monitoring System – Product Status 250
ipro 4 Glucose Monitoring System – Product Description 250
ipro 5 Glucose Monitoring System Sensor – Product Status 251
ipro 5 Glucose Monitoring System Sensor – Product Description 251
NexSensor – Product Status 251
NexSensor – Product Description 252
Simplera CGM Sensor – Product Status 252
Simplera CGM Sensor – Product Description 252
Unity Sensor – Product Status 253
Unity Sensor – Product Description 253
Medtronic Plc Pipeline Products & Ongoing Clinical Trials Overview 254
Guardian 4 Sensor – Product Status 254
Guardian 4 Sensor – Product Description 254
Medtronic Plc – Ongoing Clinical Trials Overview 255
Guardian 4 Sensor – InPen User Experience 256
Mellitor Ltd Pipeline Products & Ongoing Clinical Trials Overview 257
Mellitor Sensor – Product Status 257
Mellitor Sensor – Product Description 257
Metronom Health Inc Pipeline Products & Ongoing Clinical Trials Overview 258
CGM System – Product Status 258
CGM System – Product Description 258
MicroTech Medical Co Ltd Pipeline Products & Ongoing Clinical Trials Overview 259
AiDex G7 – Child And Adolescent Use – Product Status 259
AiDex G7 – Child And Adolescent Use – Product Description 259
AiDex X – Product Status 260
AiDex X – Product Description 260
Exactive Pro System – Product Status 260
Exactive Pro System – Product Description 261
MicroTech Medical Co Ltd – Ongoing Clinical Trials Overview 262
AiDex G7 – Child And Adolescent Use – Clinical Trial of Continuous Glucose Monitoring System Used in Pediatric Subjects with Diabetes Mellitus 263
mobiLIFE Pipeline Products & Ongoing Clinical Trials Overview 264
GlucoReader – Product Status 264
GlucoReader – Product Description 264
Mologic Ltd Pipeline Products & Ongoing Clinical Trials Overview 265
ACuTEGA – Product Status 265
ACuTEGA – Product Description 265
Movano Inc Pipeline Products & Ongoing Clinical Trials Overview 266
Movano Ring – Product Status 266
Movano Ring – Product Description 266
Nagasaki University Pipeline Products & Ongoing Clinical Trials Overview 267
Tabletop Blood-Glucose Meter – Product Status 267
Tabletop Blood-Glucose Meter – Product Description 267
Nemaura Medical Inc Pipeline Products & Ongoing Clinical Trials Overview 268
Glossary
![]()