Hemophilia A and B – Global Drug Forecast and Market Analysis, 2021-2030
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
The hemophilia A & B drug market in 8MM was valued at $9.36 billion in 2020 and is expected to grow with a CAGR of more than 4% by 2030. The key driver of growth in the hemophilia A and B markets includes rising life expectancy for hemophilia patients due to reduced morbidity and mortality associated with severe complications and reduced risk of pathogen transmission from plasma-derived sources, increasing the treated population.
The US represents the largest market in hemophilia, accounting for more than 50% of total product sales in 2020, followed by the five major European markets (5EU: France, Germany, Italy, Spain, and the UK). Recombinant factor concentrates were the leading class of drug in the eight major market (8MM) countries (the US, China, Japan, and 5EU (France, Germany, Italy, Spain, and the UK)) in 2020. Gene therapies are expected to have the highest sales in 8MM by 2030. Some of the key companies in the hemophilia A & B drug market include Roche, Novo Nordisk, Pfizer, and Sanofi.
Hemophilia A & B drug market overview
For more insights on this report, download a free report sample
What are the market dynamics of the hemophilia A & B drug market?
Major drivers of growth in the hemophilia A and B markets include the following:
Rising life expectancy for hemophilia patients, for example, due to reduced morbidity and mortality associated with severe complications and reduced risk of pathogen transmission from plasma-derived sources, increasing the treated population. The continued preference for prophylactic regimens due to proven benefits in terms of bleed rates and joint damage, supported by the availability of and switching to long-acting products and ACPs with lower dosing frequency and improved convenience.
Major barriers to growth in the hemophilia A and B markets include:
Declining prevalent cases of hemophilia, cases in Germany and Spain as well as delayed diagnosis of the disease in China. Possible market access issues for gene therapies due to their high upfront costs, lack of long-term data, and robust evidence of efficacy and safety. Limited scope for new products such as long-acting replacement factors to command a higher price point than current gold standards due to the already high cost of recombinant prophylaxis
What are the key categories of the hemophilia A & B drug market?
The hemophilia A & B drug market is categorized based on the class of drugs into DDAVP (desmopressin), plasma-derived factor concentrates, recombinant factor concentrates, monoclonal antibodies, antisense oligonucleotides, and gene therapies. Recombinant factor concentrates were the leading class of drug in 8MM, in 2020. Gene therapies is expected to have the highest sales in 8MM by 2030 with a market share of at least more than 35% in all of the major markets.
DDAVP, antisense oligonucleotide and gene therapies had the lowest sales in 2020 in 8MM, however, antisense oligonucleotide is expected to have moderate growth during the forecast period, while gene therapies are set to become the leading class of drugs by 2030.
Hemophilia A & B drug market, by key categories
For more segment insights, download a free report sample
Which are the major companies in the hemophilia A & B drug market?
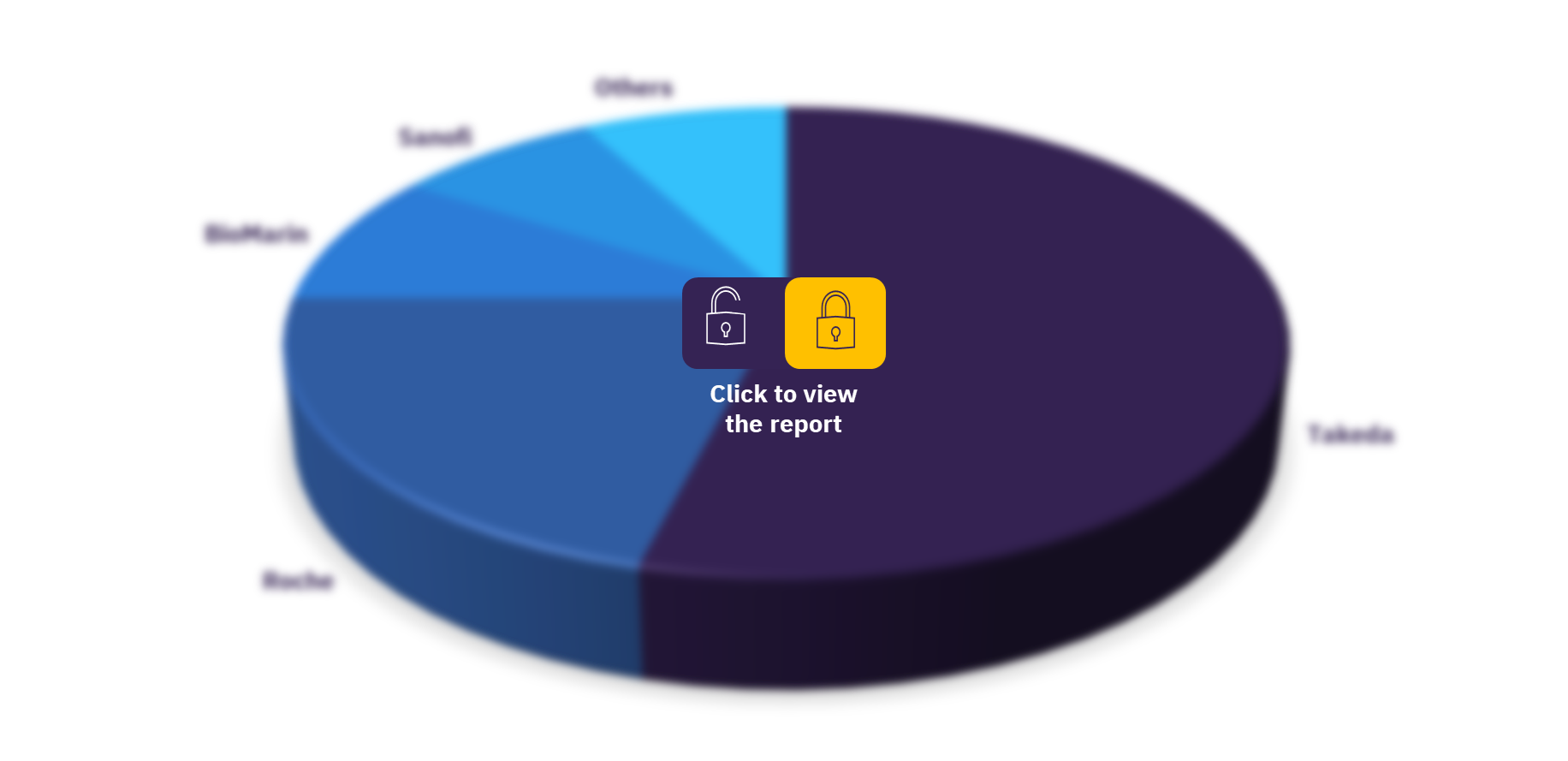
The leading companies in the hemophilia A & B drug market, include Aptevo, Bayer, BioMarin, Catalyst Biosciences, CSL Behring, GC Pharma, Kaifeng Pharmaceutical, KM Biologics, LFB, Novo Nordisk, Pfizer, Octapharma, Roche, Sanofi, SinoCelltech, Spark Therapeutics, Takeda, and UniQure. Historically, Takeda has been the dominant player in the recombinant hemophilia market with a wide range of products owned by the company including the rFVIII products Recombinate and Advate. However, the approval of Hemlibra to treat hemophilia A patients with and without inhibitors in 2018 saw Roche leapfrog Takeda to become the market leader. BioMarin is expected to become the market leader by 2030 if its gene therapy product, Roctavian, is approved for use in hemophilia A without inhibitors.
Hemophilia A & B drug market, by key players
To know more about key players, download a free report sample
Market report scope
| Market size (year – 2020) | $9.36 billion |
| Growth rate (CAGR) | >4% |
| Base year for estimation | 2020 |
| Forecast period | 2021-2030 |
| High potential countries | 8MM (the US, China, Japan, and 5EU (France, Germany, Italy, Spain, and the UK)) |
| Key categories | DDAVP (desmopressin), plasma-derived factor concentrates, recombinant factor concentrates, monoclonal antibodies, antisense oligonucleotides, and gene therapies. |
| Key companies | Aptevo, Bayer, BioMarin, Catalyst Biosciences, CSL Behring, GC Pharma, Kaifeng Pharmaceutical, KM Biologics, LFB, Novo Nordisk, Pfizer, Octapharma, Roche, Sanofi, SinoCelltech, Spark Therapeutics, Takeda, and UniQure. |
This report provides a comprehensive overview of the hemophilia A & B drug market. It also includes:
- Overview of Hemophilia A and B including epidemiology, pathophysiology, symptoms, diagnosis, and treatment guidelines.
- Topline Hemophilia A and B market revenue, the annual cost of therapy, and major pipeline product sales in the forecast period.
- Key topics: current treatment and pipeline therapies, unmet needs and opportunities, and the drivers and barriers affecting Hemophilia A and B therapeutics sales in the 8MM.
- Pipeline analysis: Comprehensive data split across different phases, emerging novel trends under development, and detailed analysis of late-stage pipeline drugs (Phase II-III).
- Analysis of the current and future market competition in the global Hemophilia A and B therapeutics market. Insightful review of the key industry drivers and challenges. Each trend is independently researched to provide a qualitative analysis of its implications.
Reasons to Buy
- Develop and design your in-licensing and out-licensing strategies, using a detailed overview of current pipeline products and technologies to identify companies with the most robust pipelines.
- Develop business strategies by understanding the trends shaping and driving the global Hemophilia A and B therapeutics market.
- Drive revenues by understanding the key trends, innovative products and technologies, market segments, and companies likely to impact the global Hemophilia A and B market in the future.
- Formulate effective sales and marketing strategies by understanding the competitive landscape and by analyzing the performance of various competitors.
- Identify emerging players with potentially strong product portfolios and create effective counterstrategies to gain a competitive advantage.
- Track drug sales in the global Hemophilia A and B therapeutics market from 2020-2030.
- Organize your sales and marketing efforts by identifying the market categories and segments that present maximum opportunities for consolidations, investments, and strategic partnerships.
Bayer
BioMarin
Catalyst Biosciences
CSL Behring
GC Pharma
Kaifeng Pharmaceutical
KM Biologics
LFB
Novo Nordisk
Pfizer
Octapharma
Roche
Sanofi
SinoCelltech
Spark Therapeutics
Takeda
UniQure
Table of Contents
Table
Figures
Frequently asked questions
-
What was the market value of the hemophilia A & B drug market in 2020?
The 8MM hemophilia A & B drug market was valued at $9.36 billion in 2020.
-
What is the growth rate of the hemophilia A & B drug market?
The hemophilia A & B drug market is expected to grow by a CAGR of more than 4% during 2021-2030.
-
What are the key categories of the hemophilia A & B drug market?
The hemophilia A & B drug market is categorized based on the class of drugs into DDAVP (desmopressin), plasma-derived factor concentrates, recombinant factor concentrates, monoclonal antibodies, antisense oligonucleotides, and gene therapies.
-
What are the key companies in the hemophilia A & B drug market?
The leading companies in the hemophilia A & B drug market are Aptevo, Bayer, BioMarin, Catalyst Biosciences, CSL Behring, GC Pharma, Kaifeng Pharmaceutical, KM Biologics, LFB, Novo Nordisk, Pfizer, Octapharma, Roche, Sanofi, SinoCelltech, Spark Therapeutics, Takeda, and UniQure.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more Hematological Disorders reports