Microbiome-Targeting Therapeutics in Infectious Diseases – Thematic Research
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
Microbiome Therapeutics Market Analysis Report Overview
Microbiome is the community of microorganisms such as fungi, bacteria, and viruses, and their genetic material existing in and on the human body. Although the primary site of aggregated microbes is in the gastrointestinal (GI) tract, populations of microorganisms are also found on the surface of the skin, in the upper and lower respiratory tract, and in the genitourinary tracts.
Microbiome-targeting drugs aim to alter the microbial composition of the gut through the administration of live microbes (probiotics), or other compounds called prebiotics, to avoid administering live bacteria. These compounds are administered to regulate and change the human microbiome composition or function beneficially. Small and mid-sized biotech companies currently dominate the pipeline for microbiome-targeting therapeutics. Players from large pharma companies, such as Takeda, have partnered with smaller biotech companies and academic labs to expedite the development of microbiome-targeting therapeutics in infectious diseases.
The microbiome-targeting therapeutics thematic intelligence report provides an overview of microbiome-targeting drugs for infectious diseases including classification of therapy and technologies, regulatory and market access details as well as product & company profiles.
Key Trends
The key trends that are shaping the microbiome theme can be classified into two categories: industry trends and regulatory trends.
- Industry Trends – No microbiome-targeting therapy has received marketing approval yet. Although regulatory agencies are adapting pre-existing guidance for other types of drugs to microbiome-targeting therapies, specific guidance for this new class of drugs will likely be established over the next decade. Challenges in the drug development process that need to be addressed include the preservation of the live microbial strains in the product until they are administered to the patient.
- Regulatory trends – In the US, microbiome-targeting therapies are classified under the category of live biotherapeutic products (LBPs), which are regulated as biologic products and include viruses, therapeutic serums, toxins, antitoxins, vaccines, blood, blood components or derivatives, allergenic products, proteins (except any chemically synthesized polypeptide), and analogous products. Despite this new regulatory designation, there are not currently any guidelines specific to New Drug Applications for LBPs.
For more insights on key trends shaping the microbiome theme, download a free report sample
Microbiome-Targeting Therapeutics in Infectious Diseases – Industry Analysis
GlobalData expects that late-stage microbiome-targeting pipeline agents will begin launching in the US as early as late 2022. Since most drugs in the pipeline are in earlier stages of clinical development, most drug launches will likely take place after 2024.
The microbiome-targeting therapeutics in infectious diseases industry analysis also covers:
- Market Outlook for Rebiotix Inc’s RBX-2660
- Market Outlook for Finch Therapeutic’s CP-101
- Market Outlook for Infant Bacterial Therapeutics’ IBP-9414
- Market Outlook for Evelo Biosciences’ EDP-1815
Microbiome-Targeting Therapeutics in Infectious Diseases - Value Chain Analysis
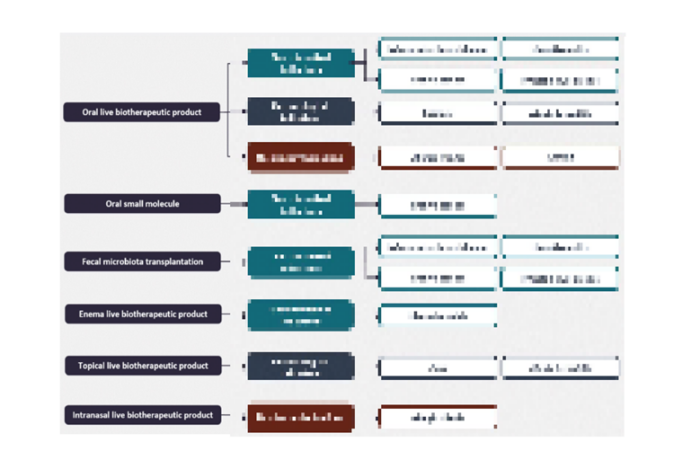
GlobalData’s microbiome value chain comprises six segments: oral live biotherapeutic product, oral small molecule, fecal microbiota transplantation, enema live biotherapeutic product, topical live biotherapeutic product, and intranasal live biotherapeutic product.
Oral LBP: Live microbes formulated into capsules for targeted intestinal delivery
Oral Small Molecule: Small molecule is taken orally to target the microbiome
Microbiome Value Chain Analysis
For more insights on the microbiome value chains, download a free report sample
Leading Companies
The leading microbiome-targeting therapeutic companies are:
- Rebiotix Inc
- Nexbiome Therapeutics
- Finch Therapeutics Group Inc
- Evelo Biosciences
- Infant Bacterial Therapeutics AB
- Osel Inc
- Mikrobiomik Healthcare Company SL
- Seres Therapeutics Inc
To know more about microbiome-targeting therapeutic companies, download a free report sample
Drug Development Thematic Scorecard – Microbiome Theme
- GlobalData’s Drug Development Thematic Scorecard provides a comprehensive outlook for players in the drug development sector over the next two to four years, based on 10 of the major themes set to transform the landscape including Microbiome.
- It includes 53 companies from across the industry, including biopharmaceutical companies, biosimilar, and generic manufacturers, contract development and manufacturing organizations, and contract research organizations.
- Using GlobalData’s core datasets, such as sales forecasts and deals, companies are scored on a scale of 1 to 5, where 1 is vulnerable and 5 is dominant.
Drug Development Thematic Scorecard
To know more about the sector scorecards, download a free report sample
Microbiome-Targeting Therapeutics Market Report Overview
| Report Pages | 59 |
| Regions Covered | Global |
| Key Trends | Industry Trends and Regulatory Trends |
| Value Chains | Oral Live Biotherapeutic Product, Oral Small Molecule, Fecal Microbiota Transplantation, Enema Live Biotherapeutic Product, Topical Live Biotherapeutic Product, and Intranasal Live Biotherapeutic Product |
| Leading Companies | Rebiotix Inc, Nexbiome Therapeutics, Finch Therapeutics Group Inc, Evelo Biosciences, Infant Bacterial Therapeutics AB, Osel Inc, Mikrobiomik Healthcare Company SL, and Seres Therapeutics Inc |
Reasons to Buy
- What impact will late-stage pipeline agents have on the market? Which class of IO drugs will have the highest peak sales, and why?
- What are the current unmet needs and which pipeline drugs or strategies are positioned to counter these unmet needs? What are the opportunities for R&D in microbiome-targeting in the ID space?
- How is the field of microbiome-targeting therapies in ID going to move forward? Which indications are the most promising for combination therapy?
- What is the regulatory landscape for live biotherapeutic products (LBP) in the US and 5EU?
- What is the current and future outlook for microbiome-targeting therapeutics according to KOLs?
Nexbiome Therapeutics
Finch Therapeutics Group
Evelo Biosciences
Infant Bacterial Therapeutics
Osel Inc
Mikrobiomik Healthcare
Seres Therapeutics
Table of Contents
Frequently asked questions
-
What are microbiomes?
Microbiomes are the communities of microorganisms such as fungi, bacteria, and viruses, and their genetic material existing in and on the human body.
-
What are the key trends impacting the microbiome-targeting therapeutics market?
The key trends that will impact the microbiome-targeting therapeutics market can be classified into two categories: industry trends and regulatory trends.
-
What are the components of the microbiome value chains?
GlobalData’s microbiome value chain comprises six segments: oral live biotherapeutic product, oral small molecule, fecal microbiota transplantation, enema live biotherapeutic product, topical live biotherapeutic product, and intranasal live biotherapeutic product.
-
Which are the leading companies in the microbiome-targeting therapeutics market for infectious diseases?
The leading companies in the microbiome-targeting therapeutics market for infectious diseases are Rebiotix Inc, Nexbiome Therapeutics, Finch Therapeutics Group Inc, Evelo Biosciences, Infant Bacterial Therapeutics AB, Osel Inc, Mikrobiomik Healthcare Company SL, and Seres Therapeutics Inc.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more Pharmaceuticals reports