Transcatheter Aortic Valve Replacement (TAVR) – Pipeline Products by Stage of Development 16
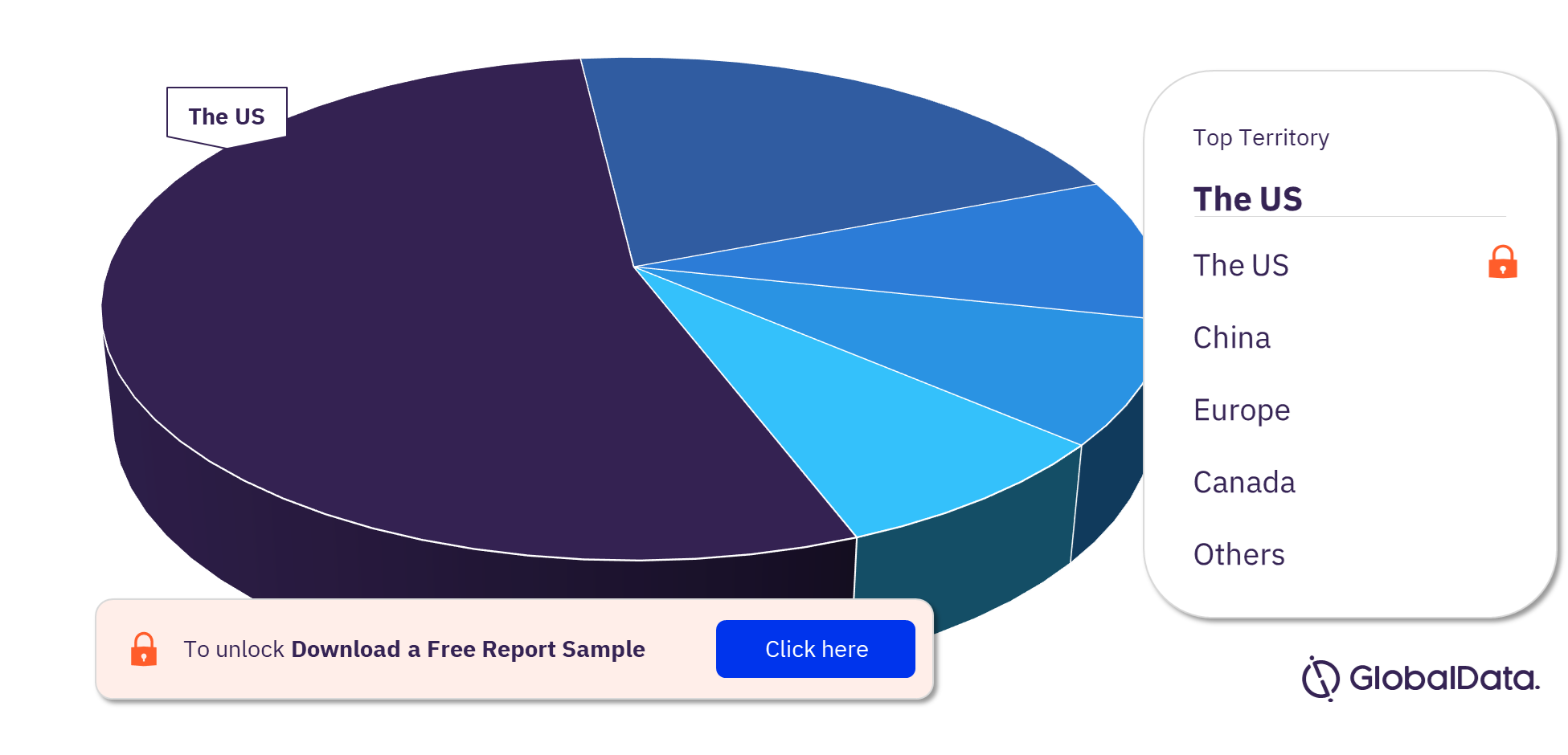
Transcatheter Aortic Valve Replacement (TAVR) – Pipeline Products by Territory 17
Transcatheter Aortic Valve Replacement (TAVR) – Pipeline Products by Regulatory Path 19
Transcatheter Aortic Valve Replacement (TAVR) – Pipeline Products by Estimated Approval Date 20
Transcatheter Aortic Valve Replacement (TAVR) – Ongoing Clinical Trials 21
Transcatheter Aortic Valve Replacement (TAVR) Companies – Pipeline Products by Stage of Development 22
Transcatheter Aortic Valve Replacement (TAVR) – Pipeline Products by Stage of Development 25
Aegis Surgical Limited Pipeline Products & Ongoing Clinical Trials Overview 28
pTAo-TAVR – Product Status 28
pTAo-TAVR – Product Description 28
Anteris Technologies Ltd Pipeline Products & Ongoing Clinical Trials Overview 29
DurAVR THV System – Product Status 29
DurAVR THV System – Product Description 29
Anteris Technologies Ltd – Ongoing Clinical Trials Overview 30
DurAVR THV System – Use of DurAVR THV System in Subjects with Severe Aortic Stenosis: First In Human Study 31
AorTx, Inc. Pipeline Products & Ongoing Clinical Trials Overview 32
AorTx Valve – Product Status 32
AorTx Valve – Product Description 32
Arbor Surgical Technologies, Inc. Pipeline Products & Ongoing Clinical Trials Overview 33
Trilogy Aortic Valve System – Product Status 33
Trilogy Aortic Valve System – Product Description 33
Atlas Medical Pipeline Products & Ongoing Clinical Trials Overview 34
Endurance Valve – Product Status 34
Endurance Valve – Product Description 34
Biotronik AG Pipeline Products & Ongoing Clinical Trials Overview 35
BIOVALVE Resheathable Transcatheter Aortic Valve – Product Status 35
BIOVALVE Resheathable Transcatheter Aortic Valve – Product Description 35
Biotronik AG – Ongoing Clinical Trials Overview 36
BIOVALVE Resheathable Transcatheter Aortic Valve – Safety and Clinical Performance of the Self-expanding Transcatheter BIOVALVE Prosthesis in Subjects with Severe Symptomatic Aortic Stenosis Suitable for Transfemoral Transcatheter Aortic Valve Implantation 37
Boston Scientific Corp Pipeline Products & Ongoing Clinical Trials Overview 38
ACURATE neo2 Transfemoral TAVR System – Product Status 38
ACURATE neo2 Transfemoral TAVR System – Product Description 39
Gen 4 Lotus Valve System – Product Status 39
Gen 4 Lotus Valve System – Product Description 39
Lotus Edge Sheath 15F – Product Status 40
Lotus Edge Sheath 15F – Product Description 40
Lotus Edge Valve System – 21mm – Product Status 40
Lotus Edge Valve System – 21mm – Product Description 41
Lotus Edge Valve System – 29mm – Product Status 41
Lotus Edge Valve System – 29mm – Product Description 42
LOTUS Mantra – Product Status 42
LOTUS Mantra – Product Description 42
Boston Scientific Corp – Ongoing Clinical Trials Overview 43
ACURATE neo2 Transfemoral TAVR System – ACURATE IDE: Transcatheter Replacement of Stenotic Aortic Valve Through Implantation of ACURATE in Subjects InDicatEd for TAVR 44
ACURATE neo2 Transfemoral TAVR System – ACURATE Neo AS Aortic Bioprosthesis for Implantation Using the ACURATE neo AS TF Transfemoral Delivery System in Patients with Severe Aortic Stenosis 44
ACURATE neo2 Transfemoral TAVR System – ACURATE neo2 Post Market Clinical Follow up Study 44
ACURATE neo2 Transfemoral TAVR System – PROVE ACURATE neo2 – Post Market Safety and Performance Surveillance in Aortic Stenosis 45
ACURATE neo2 Transfemoral TAVR System – Randomized Study Comparing Both Latest Generation Self-expanding Valves and Local Anesthesia Vs. Conscious Sedation in Transcatheter Aortic Valve Implantation 45
Carena Healthcare Ltd Pipeline Products & Ongoing Clinical Trials Overview 46
Pillar 3D Printed Replacement Heart Valve – Product Status 46
Pillar 3D Printed Replacement Heart Valve – Product Description 46
Chengdu Silara Meditech Inc Pipeline Products & Ongoing Clinical Trials Overview 47
Transcatheter Aortic Valve System – Product Status 47
Transcatheter Aortic Valve System – Product Description 47
Chengdu Silara Meditech Inc – Ongoing Clinical Trials Overview 48
Transcatheter Aortic Valve System – A Prospective, Multi-center, Single-arm Clinical Trial to Evaluate the Safety and Effectiveness of Transcatheter Aortic Valve System in Patients with Severe Aortic Stenosis with or without Moderate or Lower Degree Regurgitation 49
Transcatheter Aortic Valve System – To Evaluate Safety and Effectiveness of Transcatheter Aortic Valve System in Patients With Severe Symptomatic Aortic Insufficiency 49
Colibri Heart Valve LLC Pipeline Products & Ongoing Clinical Trials Overview 50
Colibri TAVI System – Product Status 50
Colibri TAVI System – Product Description 50
Delta Aortic Heart Valve – Product Status 51
Delta Aortic Heart Valve – Product Description 51
Second-Generation TAVI System – Product Status 51
Second-Generation TAVI System – Product Description 52
Venibri Transcatheter Heart Valve System – Product Status 52
Venibri Transcatheter Heart Valve System – Product Description 52
Colibri Heart Valve LLC – Ongoing Clinical Trials Overview 53
Colibri TAVI System – A Prospective, Single Arm Clinical Investigation Evaluating Safety and Performance of the Colibri Transcatheter Aortic Heart Valve System for the Treatment of Symptomatic Severe Aortic Stenosis Via Transfemoral Access in High Surgical Risk Patients 54
ContiTech (Beijing) Medical Technology Co Ltd Pipeline Products & Ongoing Clinical Trials Overview 55
Balloon-Expandable Transcatheter Aortic Valve Replacement Device – Product Status 55
Balloon-Expandable Transcatheter Aortic Valve Replacement Device – Product Description 55
Daidalos Solutions BV Pipeline Products & Ongoing Clinical Trials Overview 56
Sutureless Trans-Catheter Aortic Valve Replacement (TAVR) – Product Status 56
Sutureless Trans-Catheter Aortic Valve Replacement (TAVR) – Product Description 56
Direct Flow Medical Inc (Inactive) Pipeline Products & Ongoing Clinical Trials Overview 57
18F Direct Flow Medical Transcatheter Aortic Valve System – Product Status 57
18F Direct Flow Medical Transcatheter Aortic Valve System – Product Description 57
22F Percutaneous Aortic Valve System – Product Status 58
22F Percutaneous Aortic Valve System – Product Description 58
Direct Flow Medical Inc (Inactive) – Ongoing Clinical Trials Overview 59
18F Direct Flow Medical Transcatheter Aortic Valve System – SALUS Trial Transcatheter Aortic Valve Replacement System Pivotal Trial the Safety and Effectiveness of the Direct Flow Medical Tanscatheter Aortic Valve System 60
DSM Dyneema BV Pipeline Products & Ongoing Clinical Trials Overview 61
Dyneema Purity – Transcatheter Heart Valve – Product Status 61
Dyneema Purity – Transcatheter Heart Valve – Product Description 61
Dura LLC Pipeline Products & Ongoing Clinical Trials Overview 62
LowPro Valve – Product Status 62
LowPro Valve – Product Description 62
VirtuTAVR System – Product Status 63
VirtuTAVR System – Product Description 63
Edwards Lifesciences Corp Pipeline Products & Ongoing Clinical Trials Overview 64
Next Gen CENTERA – Product Status 64
Next Gen CENTERA – Product Description 64
SAPIEN 3 THV – Asymptomatic Severe Aortic Stenosis – Product Status 65
SAPIEN 3 THV – Asymptomatic Severe Aortic Stenosis – Product Description 65
SAPIEN 3 THV – Moderate Aortic Stenosis – Product Status 65
SAPIEN 3 THV – Moderate Aortic Stenosis – Product Description 66
SAPIEN X4 – Product Status 66
SAPIEN X4 – Product Description 66
Edwards Lifesciences Corp – Ongoing Clinical Trials Overview 67
SAPIEN 3 THV – Moderate Aortic Stenosis – Transcatheter Aortic Valve Replacement to Unload the Left Ventricle in Patients with Advanced Heart Failure: A Randomized Trial (TAVR UNLOAD) 68
Endoluminal Technology Research LLC Pipeline Products & Ongoing Clinical Trials Overview 69
Balloon Expandable Paniagua Heart Valve – Product Status 69
Balloon Expandable Paniagua Heart Valve – Product Description 69
Florida International University Pipeline Products & Ongoing Clinical Trials Overview 70
Percutaneous Aortic Heart Valve – Product Status 70
Percutaneous Aortic Heart Valve – Product Description 70
Folda LLC Pipeline Products & Ongoing Clinical Trials Overview 71
Foldavalve – Product Status 71
Foldavalve – Product Description 71
Foldax Inc Pipeline Products & Ongoing Clinical Trials Overview 72
Tria Transcatheter Aortic Valve Replacement Device – Product Status 72
Tria Transcatheter Aortic Valve Replacement Device – Product Description 72
HLT, Inc. Pipeline Products & Ongoing Clinical Trials Overview 73
Meridian II Transcatheter Aortic Valve Replacement System – Product Status 73
Meridian II Transcatheter Aortic Valve Replacement System – Product Description 73
HLT, Inc. – Ongoing Clinical Trials Overview 74
Meridian II Transcatheter Aortic Valve Replacement System – Study to Evaluate the Safety and Performance of HLT Meridian TAVR Valve System in the Treatment of Aortic Regurgitation 75
Meridian II Transcatheter Aortic Valve Replacement System – Study to Evaluate the Safety and Performance of HLT Meridian TAVR Valve System in the Treatment of Aortic Stenosis 75
Meridian II Transcatheter Aortic Valve Replacement System – Transfemoral Replacement of Aortic Valve with HLT Meridian Valve Early Feasibility Trial 75
Meridian II Transcatheter Aortic Valve Replacement System – Transfemoral Replacement of Aortic Valve with HLT Meridian Valve Feasibility Trial Canada 76
JC Medical, Inc. Pipeline Products & Ongoing Clinical Trials Overview 77
J-Valve TAVI System – Transapical – Product Status 77
J-Valve TAVI System – Transapical – Product Description 77
J-Valve TAVI System – Transfemoral – Product Status 78
J-Valve TAVI System – Transfemoral – Product Description 78
JC Medical, Inc. – Ongoing Clinical Trials Overview 79
J-Valve TAVI System – Transapical – Efficacy and Safety Evaluation for the Interventional Aortic Valve Bioprosthesis and Delivery System in Patients with Severe Aortic Stenosis and/or Aortic Regurgitation with Elevated Surgical Risk 80
J-Valve TAVI System – Transfemoral – J-Valve Compassionate Use Cases 81
JenaValve Technology GmbH Pipeline Products & Ongoing Clinical Trials Overview 82
JenaValve Pericardial TAVR System – Product Status 82
JenaValve Pericardial TAVR System – Product Description 82
Trilogy Heart Valve System – Product Status 83
Trilogy Heart Valve System – Product Description 83
JenaValve Technology GmbH – Ongoing Clinical Trials Overview 84
JenaValve Pericardial TAVR System – A Study to Assess Safety and Effectiveness of the JenaValve Trilogy Heart Valve System in the Treatment of High Surgical Risk Patients With Symptomatic, Severe Aortic Regurgitation (AR) 85
JenaValve Pericardial TAVR System – JENAVALVE ALIGN-AS TRIAL: Safety and Effectiveness/Performance of the Transfemoral JenaValve Pericardial TAVR System in the Treatment of Patients With Symptomatic Severe Aortic Stenosis (AS) 85
JenaValve Pericardial TAVR System – THE ALIGN-AR TRIAL: Safety and Effectiveness/Performance of the Transfemoral JenaValve Pericardial TAVR System in the Treatment of Patients with Symptomatic Severe Aortic Regurgitation (AR) 85
Trilogy Heart Valve System – Trilogy Heart Valve System for Management of Patients with Aortic Valve Disease: Patient Registry and Post-market Clinical Follow-up Study 86
Jenscare Scientific Co Ltd Pipeline Products & Ongoing Clinical Trials Overview 87
Ken-Valve – Product Status 87
Ken-Valve – Product Description 87
KenFlex – Product Status 88
KenFlex – Product Description 88
Jinshi Biotechnology (Changshu) Co Ltd Pipeline Products & Ongoing Clinical Trials Overview 89
Transcatheter Aortic Valve Replacement Device – Product Status 89
Transcatheter Aortic Valve Replacement Device – Product Description 89
KOKA Lifesciences Co Ltd Pipeline Products & Ongoing Clinical Trials Overview 90
Koka Valve – Product Status 90
Koka Valve – Product Description 90
KOKA Lifesciences Co Ltd – Ongoing Clinical Trials Overview 91
Koka Valve – A Clinical Trial of the Transcatheter Aortic Valve Implantation System With a Prospective, Multi-Center, One-Arm Approach to Evaluate the Efficacy and Safety in the Treatment of Patients With Severe Aortic Stenosis 92
Lepu Medical Technology (Beijing) Co Ltd Pipeline Products & Ongoing Clinical Trials Overview 93
Sinocrown Transcatheter Aortic Valve Replacement System – Product Status 93
Sinocrown Transcatheter Aortic Valve Replacement System – Product Description 93
Transcatheter Aortic Valve Replacement System – Product Status 94
Transcatheter Aortic Valve Replacement System – Product Description 94
Transcatheter Aortic Valve System (Balloon Dilation) – Product Status 94
Transcatheter Aortic Valve System (Balloon Dilation) – Product Description 94
Lepu Medical Technology (Beijing) Co Ltd – Ongoing Clinical Trials Overview 95
Sinocrown Transcatheter Aortic Valve Replacement System – A Prospective, Multicenter, Single-arm Clinical Trial to Evaluate Safety and Efficacy of Sinocrown Transcatheter Aortic Valve Replacement System with Accessories in Patients with Symptomatic, Calcified and Severe Aortic Stenosis 96
Mayo Clinic Pipeline Products & Ongoing Clinical Trials Overview 97
Percutaneous Aortic Valve – Product Status 97
Percutaneous Aortic Valve – Product Description 97
Medtronic Plc Pipeline Products & Ongoing Clinical Trials Overview 98
Evolut Next Gen – Product Status 98
Evolut Next Gen – Product Description 98
Meril Life Sciences Pvt Ltd Pipeline Products & Ongoing Clinical Trials Overview 99
MyVal S/Ex – Product Status 99
MyVal S/Ex – Product Description 99
MicroPort CardioFlow Medtech Corp Pipeline Products & Ongoing Clinical Trials Overview 100
VitaFlow Balloon Expandable – Product Status 100
VitaFlow Balloon Expandable – Product Description 100
VitaFlow III – Product Status 101
VitaFlow III – Product Description 101
VitaFlow Liberty – Product Status 101
VitaFlow Liberty – Product Description 102
VitaFlow Novo Generation – Product Status 102
VitaFlow Novo Generation – Product Description 102
MicroPort CardioFlow Medtech Corp – Ongoing Clinical Trials Overview 103
VitaFlow Liberty – A Prospective, Multicenter, Single-arm, Objective Performance Clinical Investigation For Evaluation of the Safety and Effectiveness of MicroPort CardioFlow VitaFlow II Transcatheter Aortic Valve System in the Treatment of Severe Aortic Valve Stenosis 104
VitaFlow Liberty – Clinical Evaluation of Safety and Efficacy of Retrievable Transcatheter Aortic Valve Implantation Device for the Treatment of Severe Aortic Regurgitation: A Prospective, Multicenter Study 104
VitaFlow Liberty – Evaluating Safety and Effectiveness/Performance of the Microport CardioFlow VitaFlow II – Transcatheter Aortic Valve System: The VITALE Study 104
MicroPort MedBot (Shanghai) Co Ltd Pipeline Products & Ongoing Clinical Trials Overview 105
TAVR Surgical Robot – Product Status 105
TAVR Surgical Robot – Product Description 105
NewMed Medical Co Ltd Pipeline Products & Ongoing Clinical Trials Overview 106
Prizvalve II TAVR Valve – Product Status 106
Prizvalve II TAVR Valve – Product Description 106
Prizvalve Pro TAVR Valve – Product Status 107
Prizvalve Pro TAVR Valve – Product Description 107
Prizvalve System – Product Status 107
Prizvalve System – Product Description 107
Starr-A TAVR Valve – Product Status 108
Starr-A TAVR Valve – Product Description 108
NewMed Medical Co Ltd – Ongoing Clinical Trials Overview 109
Prizvalve System – An Exploratory Study to Evaluate the Prizvalve System in Patients With Failing Bioprosthetic Valve 110
Prizvalve System – An Exploratory Study to Evaluate the Prizvalve System in Patients with Severe Atrioventricular Annular Calcification or Failed Prosthetic Atrioventricular Valve/ Annulus Repair 110
Prizvalve System – Evaluation of the Efficacy and Safety of the Transcatheter Aortic Valve Replacement System in Patients with Severe Aortic Valve Stenosis Disease at High Surgical Risk 110
Novogate Medical Ltd. Pipeline Products & Ongoing Clinical Trials Overview 111
Wisefix System – Transcatheter Aortic Valve Implantation – Product Status 111
Wisefix System – Transcatheter Aortic Valve Implantation – Product Description 111
NVT AG Pipeline Products & Ongoing Clinical Trials Overview 112
ALLEGRA Transfemoral TAVI System – Product Status 112
ALLEGRA Transfemoral TAVI System – Product Description 112
ALLEGRA Valve-In-Valve – Product Status 113
ALLEGRA Valve-In-Valve – Product Description 113
NVT AG – Ongoing Clinical Trials Overview 114
ALLEGRA Transfemoral TAVI System – Clinical Outcomes of the NVT ALLEGRA TAVI System TF in Failing Calcified Aortic Heart Valves in a Real-world Patient Population with Elevated Surgical Risk 115
ALLEGRA Transfemoral TAVI System – Investigation of the Safety and Performance of the NVT ALLEGRA THV System with a New Delivery System in Patients with Severe Calcified Aortic Stenosis or Failed Surgical Aortic Bioprosthesis 115
Opsens Inc Pipeline Products & Ongoing Clinical Trials Overview 116
SavvyWire – Product Status 116
SavvyWire – Product Description 116
Opsens Inc – Ongoing Clinical Trials Overview 117
SavvyWire – SAvvyWire EFficacy and SafEty in Transcatheter Aortic Valve Implantation Procedures (SAFE-TAVI) 118
P+F Products + Features GmbH Pipeline Products & Ongoing Clinical Trials Overview 119
Vienna Aortic Self-Expandable Transcatheter Valve – Product Status 119
Vienna Aortic Self-Expandable Transcatheter Valve – Product Description 119
P+F Products + Features GmbH – Ongoing Clinical Trials Overview 120
Vienna Aortic Self-Expandable Transcatheter Valve – A Two -Stage First in Human (FIH) Feasibility / Pivotal Study of the Vienna Aortic Valve SE System 121
Peca Labs Inc Pipeline Products & Ongoing Clinical Trials Overview 122
Fully Synthetic Transcatheter Valve – Product Status 122
Fully Synthetic Transcatheter Valve – Product Description 122
Peijia Medical Ltd Pipeline Products & Ongoing Clinical Trials Overview 123
Peijia Balloon Expandable Valve – Product Status 123
Peijia Balloon Expandable Valve – Product Description 123
TaurusApex Polymer Leaflets TAVR System – Product Status 124
TaurusApex Polymer Leaflets TAVR System – Product Description 124
TaurusNXT – Product Status 124
TaurusNXT – Product Description 125
Peijia Medical Ltd – Ongoing Clinical Trials Overview 126
TaurusNXT – A Multi-center, Prospective Study to Evaluate the Safety and Efficacy of TaurusNXT TAVR System for Treating Calcific Severe Aortic Stenosis 127
Pi-Cardia Ltd Pipeline Products & Ongoing Clinical Trials Overview 128
ShortCut Aortic Device – Product Status 128
ShortCut Aortic Device – Product Description 128
Pi-Cardia Ltd – Ongoing Clinical Trials Overview 129
ShortCut Aortic Device – A Prospective, Multicenter, Non-randomized, Single-arm, Open-label Clinical Study to Demonstrate the Safety and Effectiveness of the ShortCut Device (The ShortCut Study) 130
ShortCut Aortic Device – A Prospective, Multicenter, Non-randomized, Single-arm, Open-label Clinical Study to Demonstrate the Safety and Performance of the Leaflex Performer 130
PolyNova Cardiovascular Inc Pipeline Products & Ongoing Clinical Trials Overview 131
Trileaflet Polymeric Prosthetic Heart Valve – Product Status 131
Trileaflet Polymeric Prosthetic Heart Valve – Product Description 131
RegenaGraft Pipeline Products & Ongoing Clinical Trials Overview 132
Synthetic Heart Valve – Product Status 132
Synthetic Heart Valve – Product Description 132
ReValve Med Inc Pipeline Products & Ongoing Clinical Trials Overview 133
Removable Transcatheter Aortic Valve – Product Status 133
Removable Transcatheter Aortic Valve – Product Description 133
SAS Cormove Pipeline Products & Ongoing Clinical Trials Overview 134
CORMOVE Percutaneous Aortic Valve – Product Status 134
CORMOVE Percutaneous Aortic Valve – Product Description 134
Shanghai Healing Medical Instruments Co Ltd Pipeline Products & Ongoing Clinical Trials Overview 135
HanchorValve – Product Status 135
HanchorValve – Product Description 135
Transcatheter Aortic Valve Replacement Device – Product Status 136
Transcatheter Aortic Valve Replacement Device – Product Description 136
Shanghai Healing Medical Instruments Co Ltd – Ongoing Clinical Trials Overview 137
HanchorValve – A Multi-center Clinical Study of HanchorValve to Treat Patients with Severe Aortic Regurgitation 138
St. Jude Medical LLC Pipeline Products & Ongoing Clinical Trials Overview 139
Portico Next Generation Transcatheter Aortic Heart Valve – Product Status 139
Portico Next Generation Transcatheter Aortic Heart Valve – Product Description 139
St. Jude Medical LLC – Ongoing Clinical Trials Overview 140
Portico Next Generation Transcatheter Aortic Heart Valve – Evaluation of the Portico NG (Next Generation) Transcatheter Aortic Valve in High and Extreme Risk Patients with Symptomatic Severe Aortic Stenosis 141
Stony Brook University Pipeline Products & Ongoing Clinical Trials Overview 142
Next Generation TAVR Valve – Product Status 142
Next Generation TAVR Valve – Product Description 142
Strait Access Technologies (Pty) Ltd Pipeline Products & Ongoing Clinical Trials Overview 143
SAT TAVI System – Product Status 143
SAT TAVI System – Product Description 143
Symetis SA Pipeline Products & Ongoing Clinical Trials Overview 144
ACURATE neo2 Aortic Valve System – Product Status 144
ACURATE neo2 Aortic Valve System – Product Description 144
ACURATE Prime – Product Status 145
ACURATE Prime – Product Description 145
ACURATE Prime XL Transfemoral Aortic Valve System – Product Status 145
ACURATE Prime XL Transfemoral Aortic Valve System – Product Description 146
ACURATE TAo – Product Status 146
ACURATE TAo – Product Description 146
NG ACURATE – Product Status 147
NG ACURATE – Product Description 147
Symetis SA – Ongoing Clinical Trials Overview 148
ACURATE neo2 Aortic Valve System – ACURATE Neo AS Aortic Bioprosthesis for Implantation Using the ACURATE neo AS TF Transfemoral Delivery System in Patients with Severe Aortic Stenosis 149
ACURATE neo2 Aortic Valve System – Evaluation of Safety and Clinical Efficacy of Boston Scientific ACURATE NEO/TF Transcatheter Heart Valves and Comparison with Medtronic EVOLUT R/ PRO Systems in All Comers Undergoing TAVR 149
ACURATE neo2 Aortic Valve System – Project to Look for Early Discharge in Patients Undergoing TAVI With ACURATE – An International Multi-center Early Discharge TAVI Program 149
ACURATE neo2 Aortic Valve System – Prospective Evaluation Complementing Investigation with Acurate Neo Device 150
ACURATE neo2 Aortic Valve System – Prospective, Randomized, Cost-utility Analysis of Transcatheter Aortic Valve Implantation Versus Surgical Aortic Valve Replacement in Brazil 150
ACURATE Prime XL Transfemoral Aortic Valve System – A Study to Evaluate the Feasibility and Safety of the ACURATE Prime XL Aortic Valve System in Patients Indicated for TAVI: ACURATE Prime XL Human Feasibility Study 151
The University of British Columbia Pipeline Products & Ongoing Clinical Trials Overview 152
Biomaterial Heart Valve – Product Status 152
Biomaterial Heart Valve – Product Description 152
Thubrikar Aortic Valve, Inc. Pipeline Products & Ongoing Clinical Trials Overview 153
Optimum TAVI System – Product Status 153
Optimum TAVI System – Product Description 153
Thubrikar Aortic Valve, Inc. – Ongoing Clinical Trials Overview 154
Optimum TAVI System – Efficacy and Safety of Optimum TAVI System in Subjects with Severe Aortic Stenosis: A First-in-Human Study 155
University College London Pipeline Products & Ongoing Clinical Trials Overview 156
TRISKELE Aortic Valve – Product Status 156
TRISKELE Aortic Valve – Product Description 156
University of California San Francisco Pipeline Products & Ongoing Clinical Trials Overview 157
Transcatheter Aortic Valve – Product Status 157
Transcatheter Aortic Valve – Product Description 157
University of Cambridge Pipeline Products & Ongoing Clinical Trials Overview 158
Removable Transcatheter Valve – Product Status 158
Removable Transcatheter Valve – Product Description 158
University of Iowa Pipeline Products & Ongoing Clinical Trials Overview 159
Cellulose Based Aortic Heart Valve – Product Status 159
Cellulose Based Aortic Heart Valve – Product Description 159
University of Pennsylvania Pipeline Products & Ongoing Clinical Trials Overview 160
Aortic Repair Device – Product Status 160
Aortic Repair Device – Product Description 160
Vascular Concepts Ltd Pipeline Products & Ongoing Clinical Trials Overview 161
TAVI (Transcatheter Aortic Valve Replacement) – Product Status 161
TAVI (Transcatheter Aortic Valve Replacement) – Product Description 161
Vascular Innovations Co Ltd Pipeline Products & Ongoing Clinical Trials Overview 162
Hydra TAVI Bioprosthesis – Product Status 162
Hydra TAVI Bioprosthesis – Product Description 162
Venus Medtech Hangzhou Inc Pipeline Products & Ongoing Clinical Trials Overview 163
AR Valve – Product Status 163
AR Valve – Product Description 164
Transapical Trinity Aortic Valve Implantation System – Product Status 164
Transapical Trinity Aortic Valve Implantation System – Product Description 164
Transfemoral Trinity Aortic Valve Implantation System – Product Status 165
Transfemoral Trinity Aortic Valve Implantation System – Product Description 165
Venus PowerX Valve – Product Status 165
Venus PowerX Valve – Product Description 166
Venus Vitae Valve – Product Status 166
Venus Vitae Valve – Product Description 166
VenusA-Plus – Product Status 167
VenusA-Plus – Product Description 167
VenusA-Pro Valve – Product Status 167
VenusA-Pro Valve – Product Description 168
VenusA-Valve – Product Status 168
VenusA-Valve – Product Description 168
Venus Medtech Hangzhou Inc – Ongoing Clinical Trials Overview 169
VenusA-Valve – Venus Panorama II Post-marketing Registry Study 170
Venus Vitae Valve – A First-in-man Clinical Trial of Venus Vitae Valve in Patients with Heart Valvular 171
Disease 171
Xeltis AG Pipeline Products & Ongoing Clinical Trials Overview 172
Transcatheter Aortic Heart Valve – Product Status 172
Transcatheter Aortic Heart Valve – Product Description 172
Youngheartvalve LLC Pipeline Products & Ongoing Clinical Trials Overview 173
Polymeric Trans-Catheter Aortic Valve – Product Status 173
Polymeric Trans-Catheter Aortic Valve – Product Description 173
Glossary 196
![]()