Vaccine Market Size and Trends Analysis by Vaccine Type, Marketed and Pipeline Products, Clinical Trials, Unmet Needs, Competitive Landscape and Forecast to 2027
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
Vaccine Market Report Overview
The vaccine market is valued at over $100 billion as of November 2021. The subunit vaccine market is projected to be the most valuable category during the forecast period, followed by conjugate vaccines. COVID-19 has had the single biggest impact in the history of the vaccine market. It has led to large amounts of investment in vaccine development, a record number of clinical trials, and academic research and collaborations. The pandemic has also led to new companies as well established companies coming up with innovative technologies in the vaccines market. The development and authorization of mRNA-based vaccines in response to the COVID-19 pandemic marks a critical milestone for scientific research and public health. The vaccine market research report provides an overview of the global vaccines market including marketed products, pipeline products, clinical trials, unmet needs, and market revenue by vaccine type.
Vaccine Market Trends
Vaccines typically have lower margins relative to therapeutics targeting oncology and neurology indications. However, spurred by advancements in recombinant technologies and nucleic acid vaccines, novel platforms have stimulated renewed interest in vaccines and validated the decision for smaller players to forge ahead in this space.
The development of vaccines and therapeutics for COVID-19 will encourage greater collaborations between academia and the industry, allowing an increased exchange of R&D findings. Academic researchers put forth a record number of COVID-19-related publications. Increased research alongside joint efforts between big pharma and smaller biotech start-ups, often in collaboration with academic institutes, has allowed for the launch of record numbers of clinical trials targeting COVID-19.
To know more about the vaccines market trends, download a free report sample
Vaccine Market Segmentation by Categories
The key vaccines in the vaccine market are Live Attenuated Vaccines, Inactivated Vaccines, Subunit Vaccines, DNA Vaccines, RNA Vaccines, Virus-Like Particles, Recombinant Vectored Vaccines, and Toxoid Vaccines.
Live Attenuated Vaccines: Live-attenuated vaccines consist of live, modified, or weakened disease-causing pathogens that produce attenuated, or limited, infection. This infection can invoke an immune response but is not severe or long-lived enough to cause a disease state.
Inactivated Vaccines: Inactivated vaccines utilize radiation, heat, or chemical reagents to inactivate the pathogen so that it cannot replicate in the host. They are unable to cause disease and are safe to use in immunosuppressed patients but may be less immunogenic than live attenuated vaccines.
Subunit Vaccines: Subunit vaccines are simplified from traditional vaccines, containing only the protein antigen subunits necessary to stimulate immunity and prevent infection. They are unable to cause infection and are safe to use in immunosuppressed patients.
To know more about the vaccines market categories, download a free report sample
Vaccine Market Analysis by Clinical Trials
Most of the pipeline drugs are in Phase I and just over half of the clinical trials with pipeline candidates are in completed status. Of the top 10 indications studied in clinical trials, COVID-19 dominates with Modern 219 studies. Industry sponsors lead the clinical trials landscape for pipeline candidates and hold trials. The National Institute of Allergy and Infectious Diseases (NIAID) sponsored the highest number of clinical trials utilizing pipeline vaccine candidates. The remainder of the top 10 sponsors are established big pharma players such as GSK, Pfizer, and Johnson & Johnson.
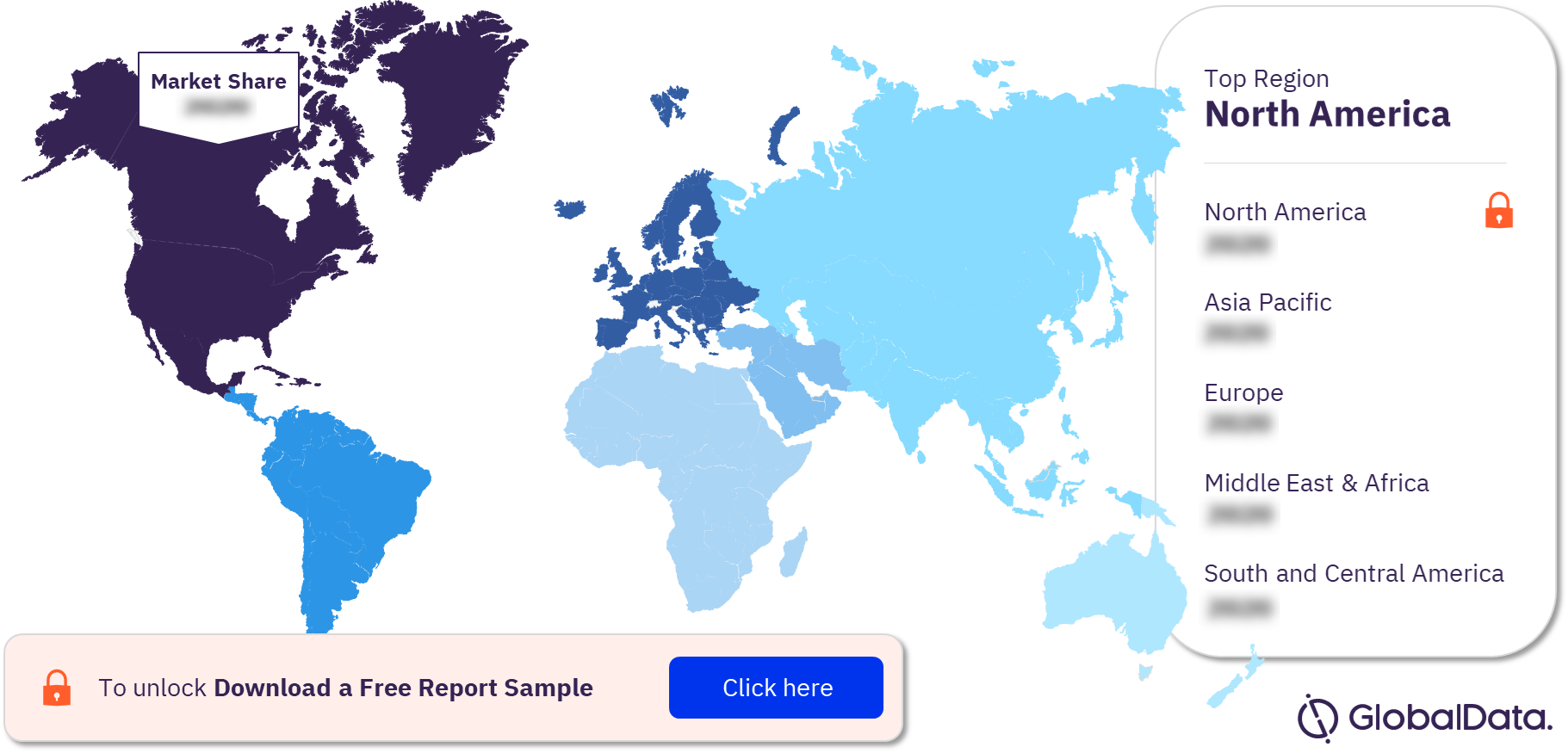
The key regions for vaccine clinical trials are North America, Asia-Pacific, Europe, the Middle East and Africa, and South and Central America. Majority of the clinical trial site locations are based in North America, followed by Asia-Pacific.
Vaccine Market Analysis by Clinical Trials
Most of the pipeline drugs are in Phase I and just over half of the clinical trials with pipeline candidates are in completed status. Of the top 10 indications studied in clinical trials, COVID-19 dominates with Modern 219 studies. Industry sponsors lead the clinical trials landscape for pipeline candidates and hold trials. The National Institute of Allergy and Infectious Diseases (NIAID) sponsored the highest number of clinical trials utilizing pipeline vaccine candidates. The remainder of the top 10 sponsors are established big pharma players such as GSK, Pfizer, and Johnson & Johnson.
The key regions for vaccine clinical trials are North America, Asia-Pacific, Europe, the Middle East and Africa, and South and Central America. Majority of the clinical trial site locations are based in North America, followed by Asia-Pacific.
Vaccine Market Analysis by Regions, 2021 (%)
To know more about the regions in the vaccines market, download a free report sample
Vaccine Market Analysis by Marketed Products
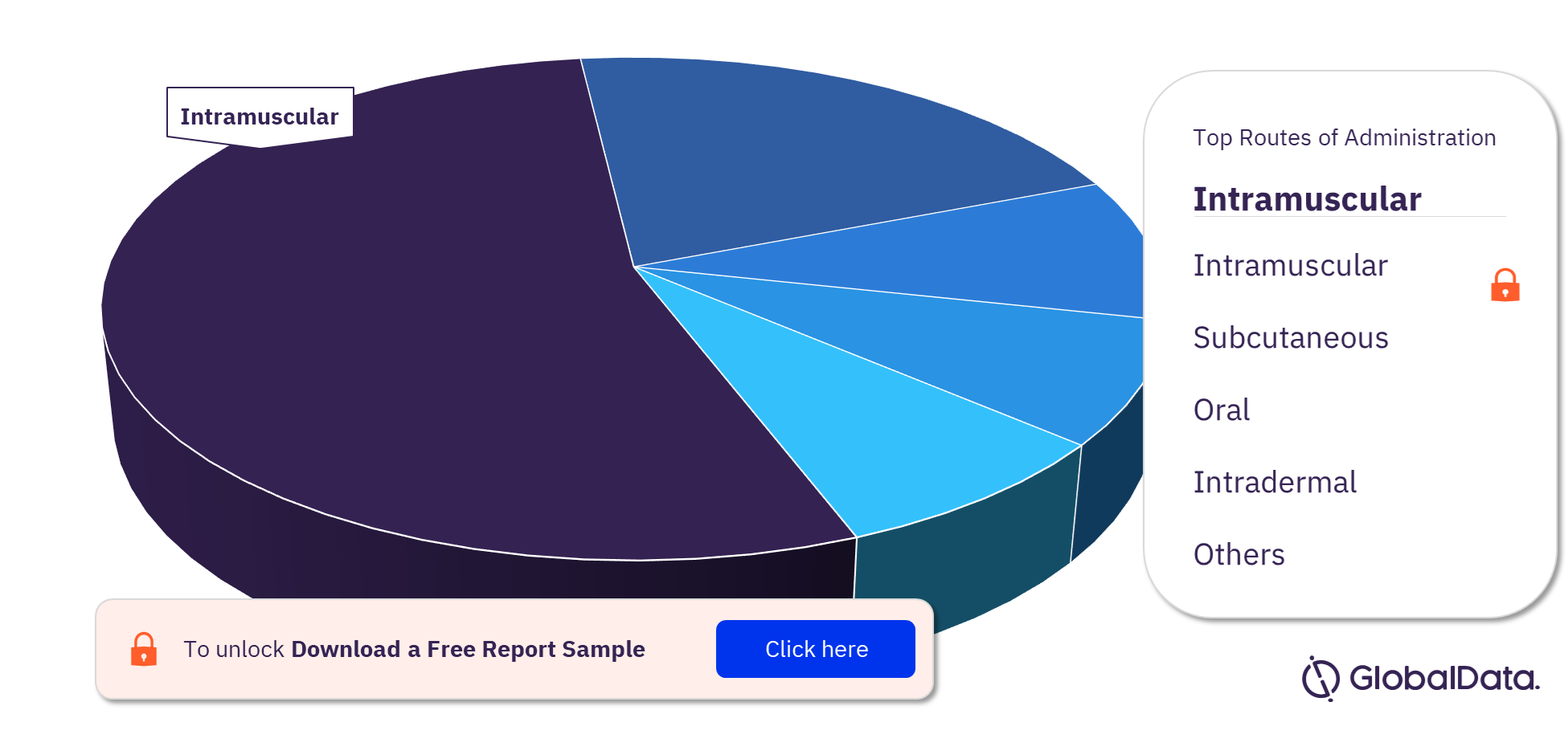
There are currently 576 marketed vaccine products including Inactivated Vaccine, Live Attenuated Vaccine, and Toxoid Vaccine. Inactivated vaccines make up the largest segment of the global marketed vaccines landscape. Most studies focus on Influenza A. The key routes of administration in the vaccine market are intramuscular, subcutaneous, oral, intradermal, nasal, intravenous, inhalational, cutaneous, and parenteral. Intramuscular administration is the most frequently used route for marketed vaccines. Typically, vaccines containing adjuvants are administered intramuscularly to circumvent a variety of side effects including irritation and skin discoloration.
Vaccine Marketed Products Analysis, by Routes of Administration, 2021 (%)
To know more about the routes of administration in the vaccines market, download a free report sample
Vaccine Market Analysis by Pipeline Products
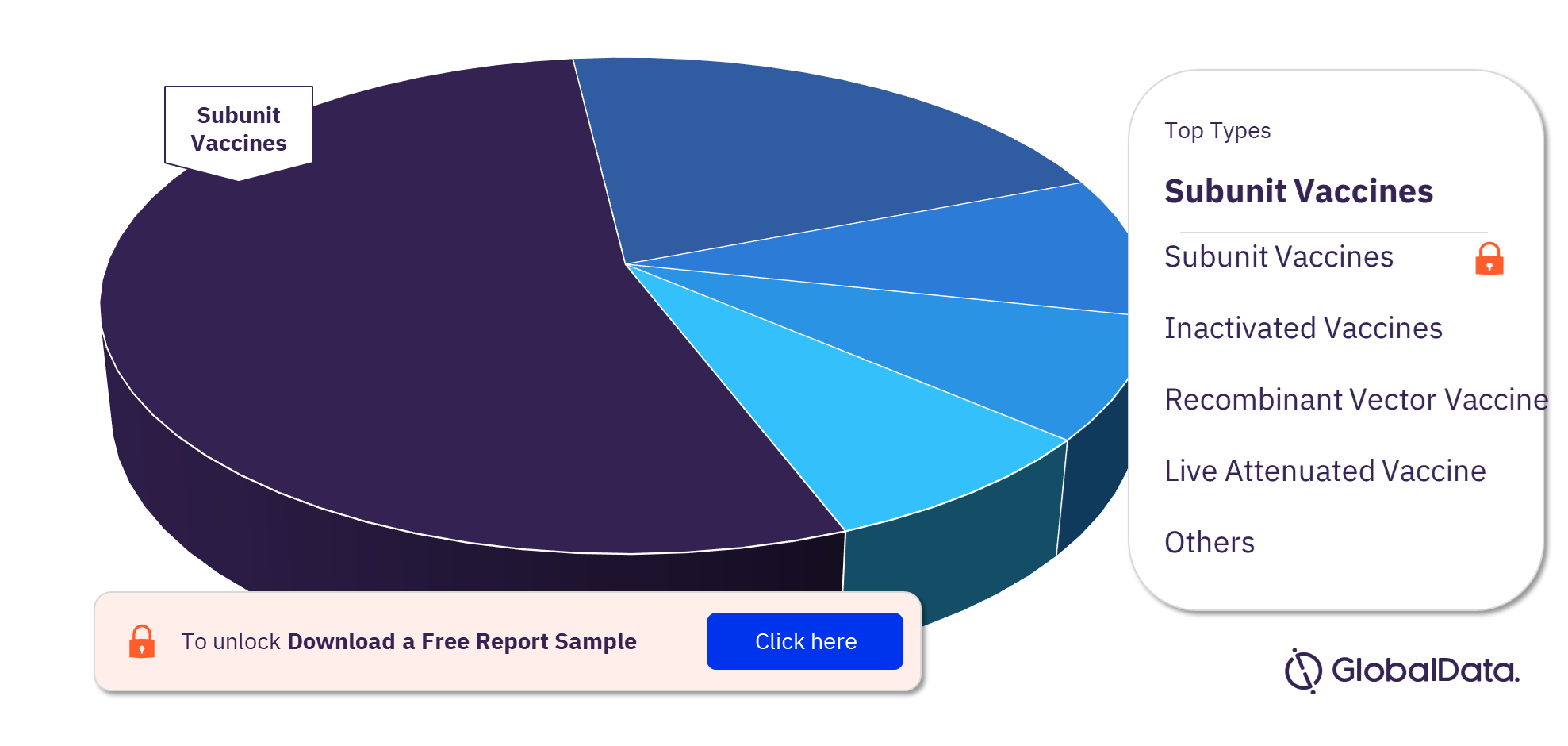
There are currently almost 700 pipeline vaccine candidates globally including subunit vaccines, inactivated vaccines, recombinant vector vaccine, live attenuated vaccine, and conjugate vaccine. Most products in development are subunit vaccines. Vaccine pipeline products by indication include Coronavirus disease (COVID-19), Influenza A virus, and Human immunodeficiency virus (HIV). Most pipeline products are in Phase I, followed by Phase II.
Vaccine Pipeline Products Analysis, by Types, 2021 (%)
To know more about the types in the vaccines market, download a free report sample
Vaccine Market - Competitive Landscape
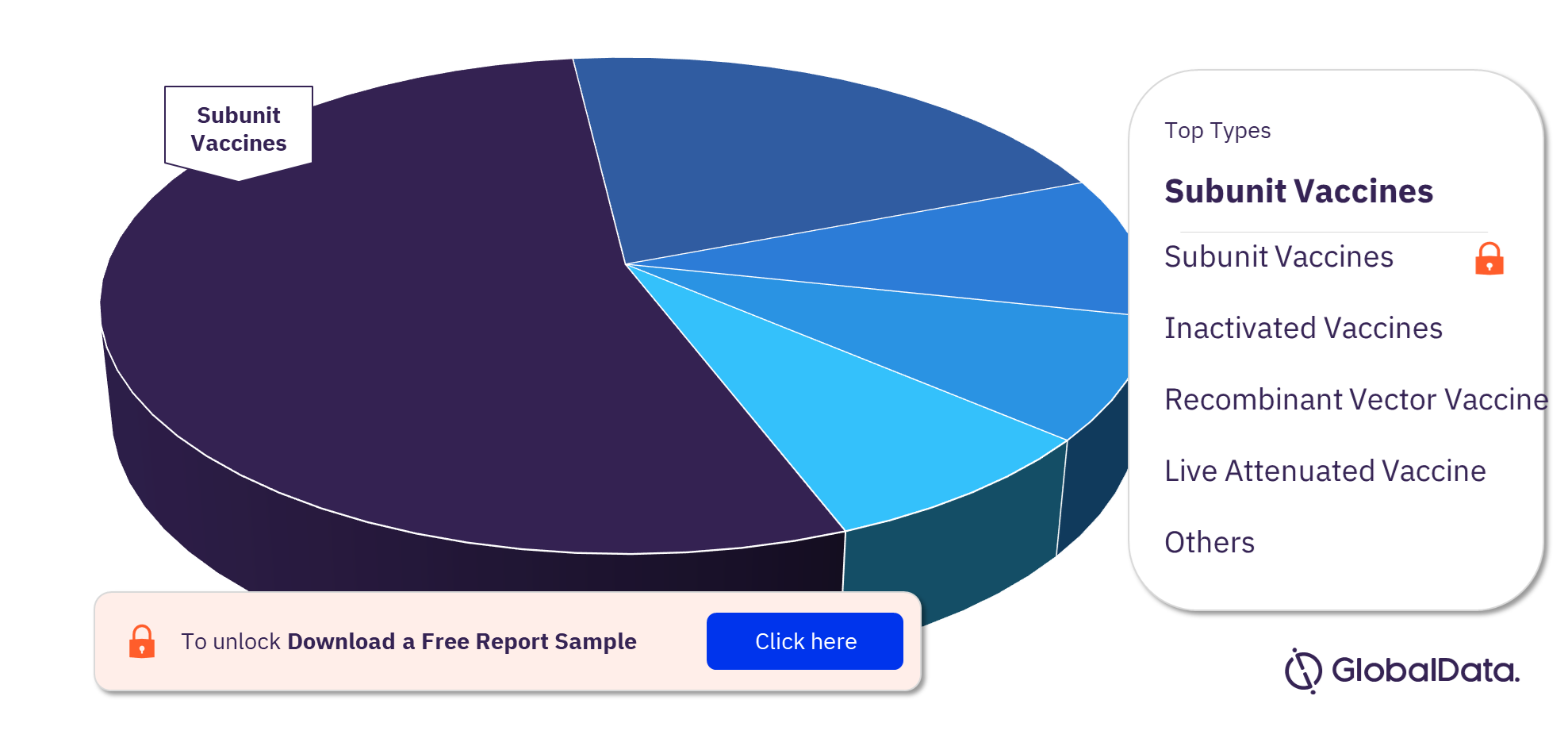
There are currently almost 700 pipeline vaccine candidates globally including subunit vaccines, inactivated vaccines, recombinant vector vaccine, live attenuated vaccine, and conjugate vaccine. Most products in development are subunit vaccines. Vaccine pipeline products by indication include Coronavirus disease (COVID-19), Influenza A virus, and Human immunodeficiency virus (HIV). Most pipeline products are in Phase I, followed by Phase II.
Vaccine Pipeline Products Analysis, by Types, 2021 (%)
To know more about the types in the vaccines market, download a free report sample
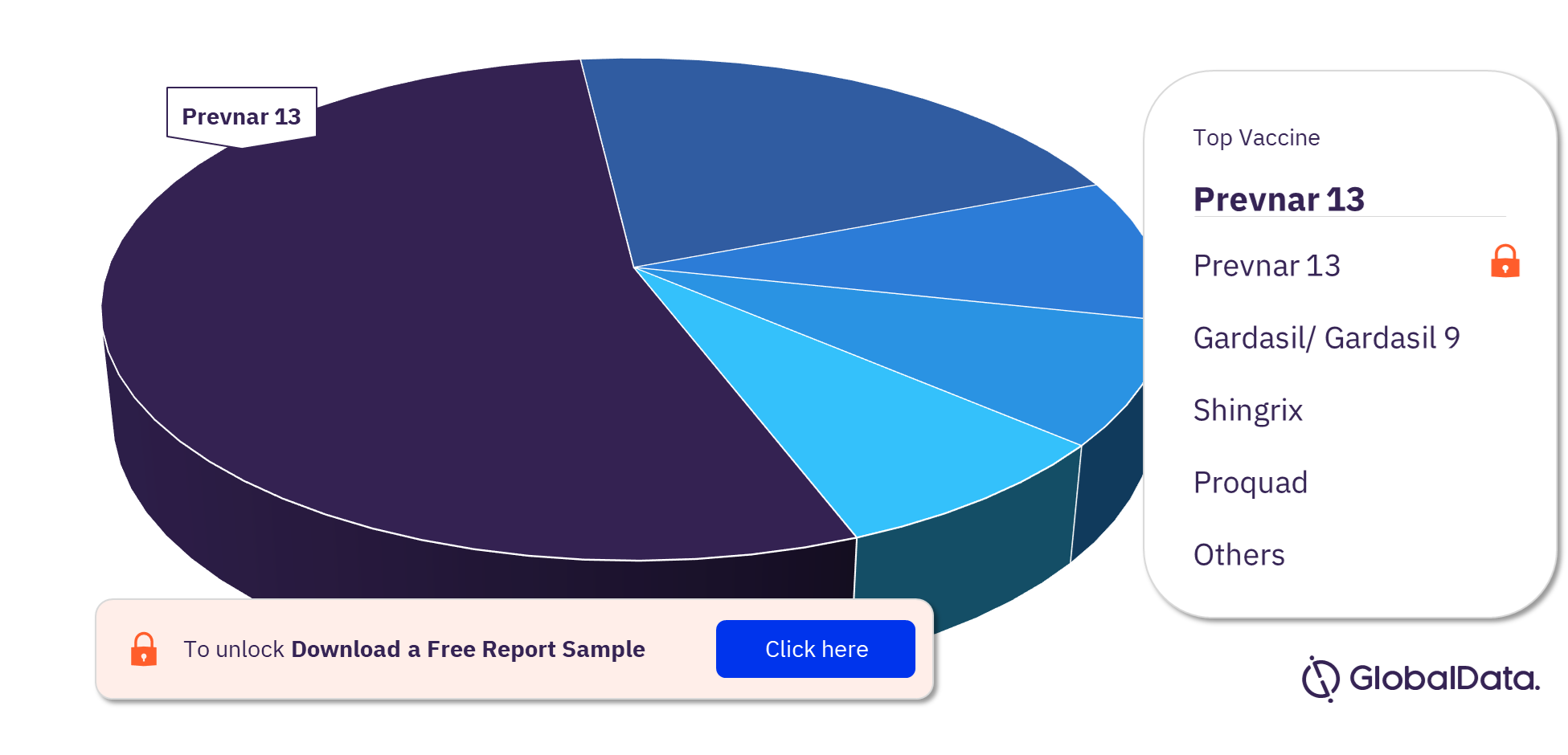
Vaccine Market - Competitive Landscape
The leading vaccines by sale in the market are Prevnar 13, Gardasil/ Gardasil 9, Shingrix, Proquad, M-M-R Ii And Varivax, Pneumovax 23, Fluarix, FluLaval, Bexsero, Infanrix, Pediarix, Rotateq, and Rotarix. Pfizer’s pneumococcal vaccine Prevnar 13 held first place in vaccine sales in 2020. It has proven to be a reliable revenue generator for the company.
Vaccine Market Analysis by Revenue, 2020 (%)
To know more about the vaccines in the vaccines market, download a free report sample
Vaccines Market Report Overview
| Key Categories | Live Attenuated Vaccines, Inactivated Vaccines, Subunit Vaccines, DNA Vaccines, RNA Vaccines, Virus-Like Particles, Recombinant Vectored Vaccines, and Toxoid Vaccines |
| Key Regions | North America, Asia-Pacific, Europe, the Middle East and Africa, and South and Central America |
| Key Routes of Administration | Intramuscular, Subcutaneous, Oral, Intradermal, Nasal, Intravenous, Inhalational, Cutaneous, and Parenteral |
| Leading Vaccines | Prevnar 13, Gardasil/ Gardasil 9, Shingrix, Proquad, M-M-R Ii And Varivax, Pneumovax 23, Fluarix, FluLaval, Bexsero, Infanrix, Pediarix, Rotateq, and Rotarix |
Segments Covered in the Report
Vaccines Categories Outlook (Value, $ Billion, 2020-2027)
- Live Attenuated Vaccines
- Inactivated Vaccines
- Subunit Vaccines
- DNA Vaccines
- RNA Vaccines
- Virus-Like Particles
- Recombinant Vectored Vaccines
- Toxoid Vaccines
Vaccines Regional Outlook (Value, $ Billion, 2020-2027)
- North America
- Asia-Pacific
- Europe
- The Middle East and Africa
- South and Central America
Scope
This report provides:
- Overview of vaccines including vaccine categories and history of vaccine development
- Overview of industry trends including the COVID-19 vaccine landscape
- Analysis of clinical trials landscape by phase of development, status, indication, trial location, and 10 top sponsors
- Analysis of marketed products by vaccine type, indication, and route of administration
- Analysis of pipeline products by vaccine type, phase of development, and indication
- Market analysis including top 10 vaccines by revenue in 2020, top 10 deals in the vaccines space, and revenue projections for 2020-2027 for subunit vaccines, conjugate vaccines, mRNA infectious disease vaccines, live attenuated vaccines, inactivated vaccines, toxoid vaccines, and DNA vaccines
- Opportunities and challenges within the HIV, malaria, Group B Streptococcus, and influenza vaccine markets
Reasons to Buy
- Understand the trends shaping and driving the global vaccines market
- Identify the innovative technologies and key players dominating the R&D landscape
- Determine which products have attracted the largest deal values
- Recognize gaps and areas of unmet need within the vaccines market
- Track sales for key vaccine subtypes from 2020-2027
Moderna
Pfizer
Johnson & Johnson
BioNTech SE
Takeda (Pty) Ltd
CanSino Biologics Inc
Gamaleya Federal Research Center of Epidemiology and Microbiology
Sinopharm Group Co Ltd
Bharat Biotech Ltd
Zydus Lifesciences Ltd
Chongqing Zhifei Biological Products Co Ltd
Sinovac Biotech Ltd
Oxford University
Novavax Inc
Curevac NV
GSK plc
AnGes Inc
Akston Biosciences Corp
Sanofi
Merck & Co Inc
Novartis AG
Ligand Pharmaceuticals Inc
Shantha Biotech
Table of Contents
Frequently asked questions
-
What was the vaccines market size in 2021?
The vaccine market is valued at over $100 billion as of November 2021.
-
What are the key vaccines in the vaccine market?
The key vaccines in the vaccine market are Live Attenuated Vaccines, Inactivated Vaccines, Subunit Vaccines, DNA Vaccines, RNA Vaccines, Virus-Like Particles, Recombinant Vectored Vaccines, and Toxoid Vaccines.
-
What are the key regions in the vaccine market?
The key regions for vaccine clinical trials are North America, Asia-Pacific, Europe, the Middle East and Africa, and South and Central America.
-
What are the key routes of administration in the vaccine market?
The key routes of administration in the vaccine market are intramuscular, subcutaneous, oral, intradermal, nasal, intravenous, inhalational, cutaneous, and parenteral.
-
Which are the leading vaccines by sale?
The leading vaccines by sale in the market are Prevnar 13, Gardasil/ Gardasil 9, Shingrix, Proquad, M-M-R Ii And Varivax, Pneumovax 23, Fluarix, FluLaval, Bexsero, Infanrix, Pediarix, Rotateq, and Rotarix.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more Vaccine reports