Clot Management Devices Pipeline by Development Stages, Segments, Region and Countries, Regulatory Path and Key Companies
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
Clot Management Devices Pipeline Market Report Overview
Clot Management Devices are used for the removal or dissolution of a clot from a blood vessel to restore proper blood circulation. This device is used as a routine procedure while performing percutaneous coronary intervention and for treating potentially life-threatening complications.
The clot management devices pipeline market research report provides comprehensive information on the products with their comparative segment-wise analysis in various stages of development. The report also covers the regional and country-wise clot management devices’ clinical trials outlook. The regulatory paths followed for conducting the clot management devices pipeline research are extensively highlighted in the report. Leading companies involved in the clot management devices pipeline development are also enlisted in this research report for better understanding of the competitive landscape.
| Key Segments | · Thrombectomy Systems (Catheters)
· Retrievable IVCF · Inferior Vena Cava Filters (IVCF) · Embolectomy Balloon Catheters · Manual Aspiration Thrombectomy Devices · Permanent IVCF · IVCF Retrieval Devices |
| Key Territories | · The US
· China · Japan · Europe · Mexico |
| Key Regulatory Paths | · 510(k)
· NMPA · Ninsho · ANVISA · CE |
| Leading Companies | · AorticLab Sarl
· APT Medical Inc · Boston Scientific Corp · Caeli Vascular LLC · Capture Vascular Inc · Embolx, Inc. · Inari Medical Inc |
| Enquire & Decide | Discover the perfect solution for your business needs. Enquire now and let us help you make an informed decision before making a purchase. |
Clot Management Devices Pipeline Market by Segments
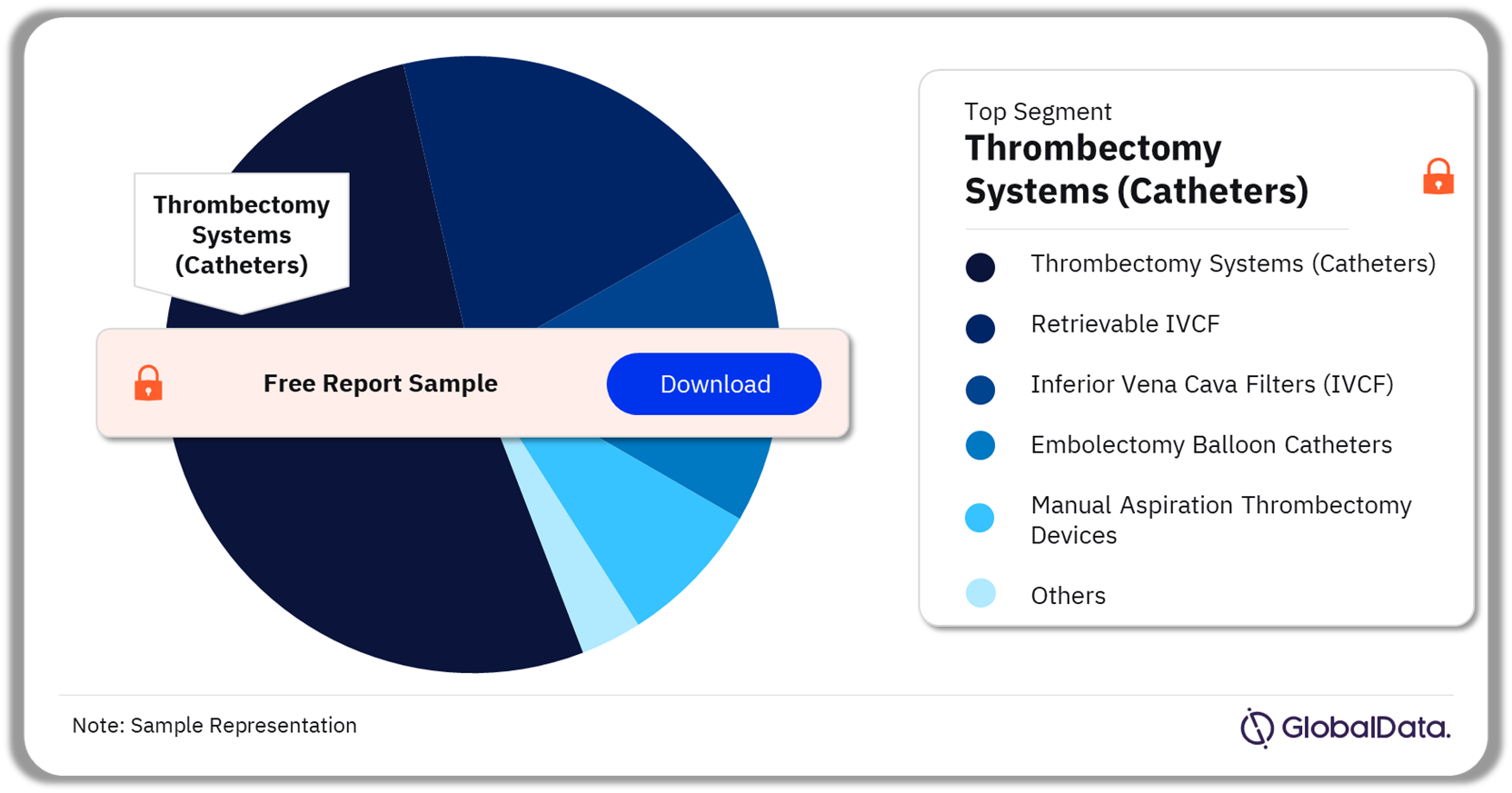
The key segments in the clot management devices pipeline market are Thrombectomy Systems (Catheters), Retrievable IVCF, Inferior Vena Cava Filters (IVCF), Embolectomy Balloon Catheters, Manual Aspiration Thrombectomy Devices, Permanent IVCF, and IVCF Retrieval Devices. As of February 2024, the Thrombectomy Systems (Catheters) segment held the highest number of pipeline products.
Clot Management Devices Pipeline Market Analysis by Segments, 2024 (%)
Buy the Full Report for Clot Management Devices Pipeline Market Segment Insights
Clot Management Devices Pipeline Market Segmentation by Territories
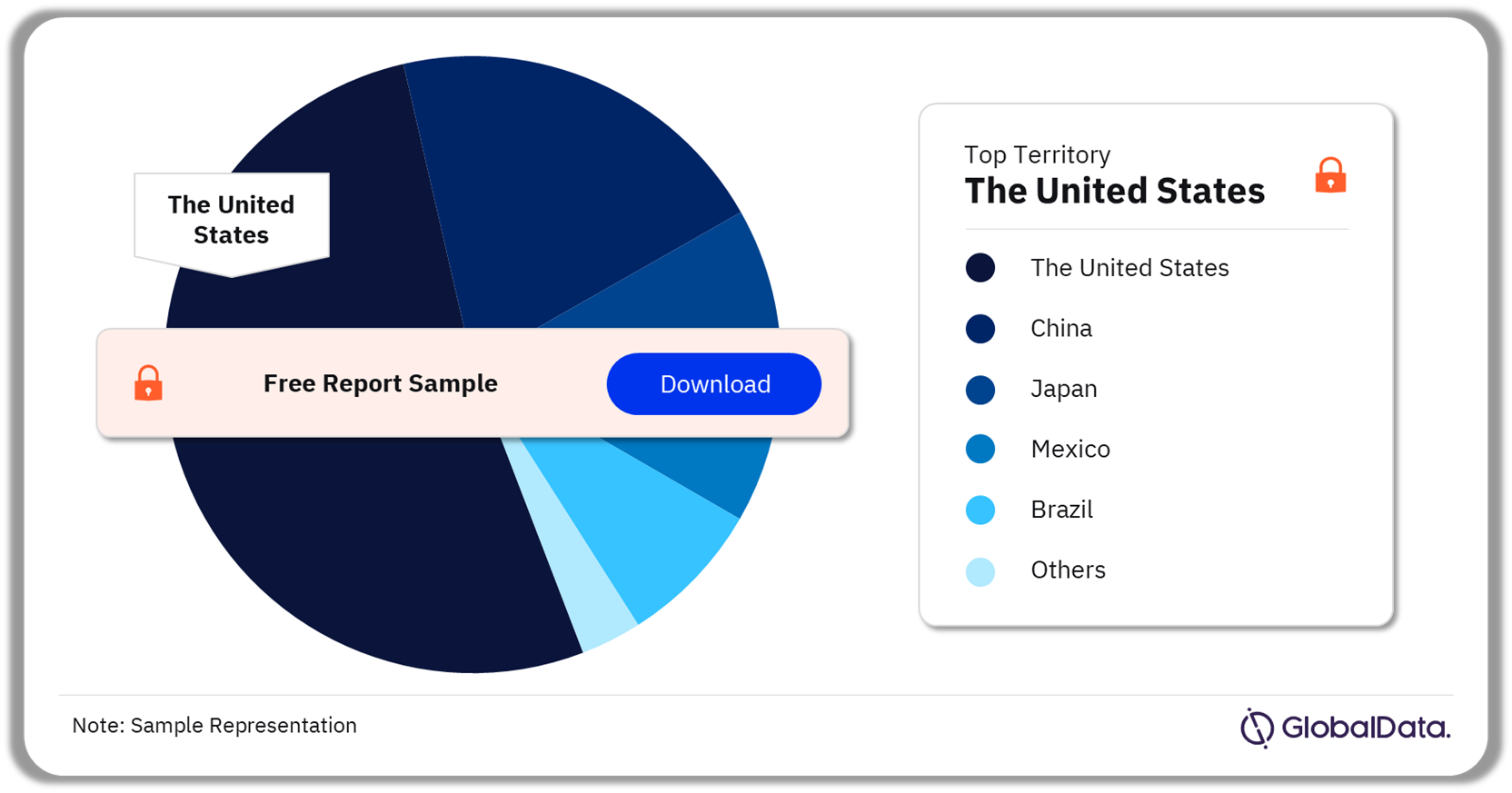
The key territories in the clot management devices market are the United States, China, Japan, Mexico, and Europe among others. As of February 2024, the United States accounted for the highest number of clot management devices in the development pipeline.
Clot Management Devices Pipeline Market Analysis by Territories, 2024 (%)
Buy the Full Report for Territorial Clot Management Devices Pipeline Market Insights
Clot Management Devices Pipeline Market Segmentation by Regulatory Paths
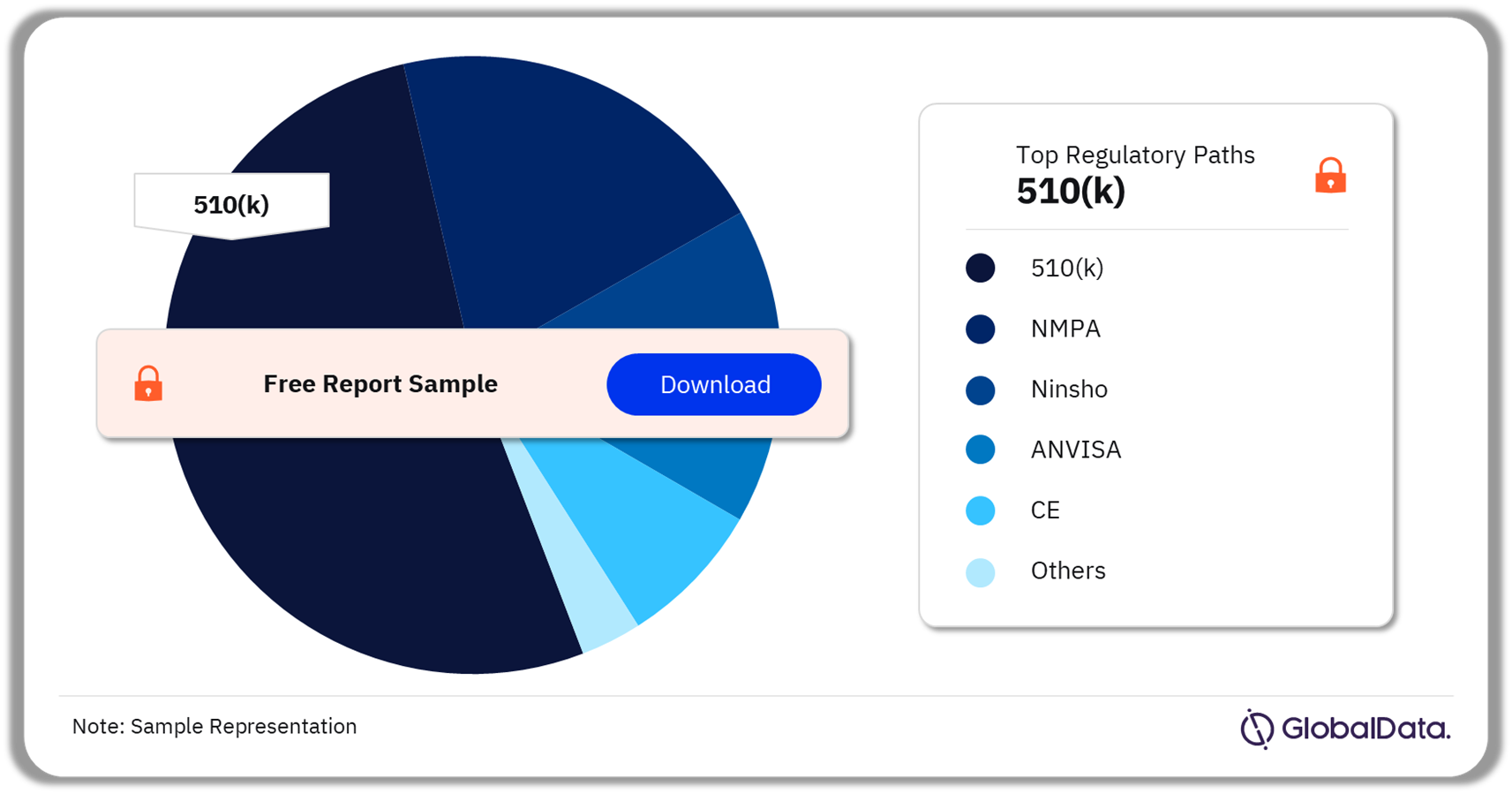
The key regulatory paths in the clot management devices pipeline market are 510(k), NMPA, Ninsho, ANVISA, and CE among others. As of February 2024, 510(k) was the most followed pathway for pipeline products in the market.
Clot Management Devices Pipeline Market Analysis by Regulatory Paths, 2024 (%)
Buy the Full Report for More Regulatory Path Insights into the Clot Management Devices Pipeline Market
Download A Free Report Sample
Clot Management Devices Pipeline Market – Competitive Landscape
The companies in the clot management devices pipeline market are:
- AorticLab Sarl
- APT Medical Inc
- Boston Scientific Corp
- Caeli Vascular LLC
- Capture Vascular Inc
- Embolx, Inc.
- Inari Medical Inc.
AorticLab Sarl: AorticLab Sarl, headquartered in Italy, provides medical and technological innovations developing next-generation devices for safe and effective transcatheter treatment of the stenotic aortic native valve. The company offers GEMINI, a Transcatheter Debridement Device (TDD) intended for peripheral artery disease management. It is designed to break and fragment the calcific plaques present on the aortic and bioprosthetic leaflets. It is obtained by double ultrasound frequency treatment generating a cavitation effect inside the leaflet tissue.
Clot Management Devices Pipeline Market Analysis by Companies, 2024
Buy the Full Report for the Leading Clot Management Devices Pipeline Development Companies
Download A Free Report Sample
Segments Covered in the Report
Clot Management Devices Pipeline Market Segments Outlook
- Thrombectomy Systems (Catheters)
- Retrievable IVCF
- Inferior Vena Cava Filters (IVCF)
- Embolectomy Balloon Catheters
- Manual Aspiration Thrombectomy Devices
- Permanent IVCF
- IVCF Retrieval Devices
Clot Management Devices Pipeline Market Territories Outlook
- The US
- Europe
- China
- Japan
- Mexico
Clot Management Devices Pipeline Market Regulatory Paths Outlook
- 510(k)
- NMPA
- CE
- MDITAC
- Ninsho
Scope
This report provides:
- Extensive coverage of the clot management devices under development.
- Details of major pipeline products which include product description, licensing and collaboration details and other developmental activities.
- Reviews of major players involved in the development of clot management devices and lists all their pipeline projects.
- Coverage of pipeline products based on various stages of development ranging from early development to approved/issued stage.
- Provides key clinical trial data related to ongoing clinical trials specific to pipeline products.
- Recent developments in the segment/industry.
Reasons to Buy
- Formulate significant competitor information, analysis, and insights to improve R&D strategies.
- Identify emerging players with potentially strong product portfolio and create effective counter strategies to gain a competitive advantage.
- Identify and understand important and diverse types of clot management devices under development.
- Develop market-entry and market-expansion strategies.
- Plan mergers and acquisitions effectively by identifying major players with the most promising pipeline.
- In-depth analysis of the product’s current stage of development, territory, and estimated launch date.
Adient Medical Inc.
Akura Medical Inc
AngioDynamics Inc
AorticLab Sarl
APT Medical Inc
Boston Scientific Corp
Caeli Vascular LLC
Capture Vascular Inc
Contego Medical LLC
CyndRX LLC
Embolx, Inc.
Endovascular Engineering Inc
Flow Medical Solutions Inc
Inari Medical Inc
Innova Vascular Inc
Interventional & Surgical Innovations, LLC
Jiangsu Zhenyi Medical Technology Co Ltd
Kaleidoscope
Koninklijke Philips NV
Lazarus Effect, Inc
LeMaitre Vascular Inc
Liquet Medical Inc
Lithotech Medical Ltd
Magneto Thrombectomy Solutions Ltd
Merit Medical Systems Inc
MicroPort Scientific Corp
Morningside (Nantong) Medical Co Ltd
NexGen Medical Systems Inc
OrbusNeich
OsciFlex LLC
Penumbra Inc
Shanghai Changde Medical Technology Co Ltd
Shanghai HeartCare Medical Technology Co Ltd
Shanghai MicroPort Endovascular MedTech Group Co Ltd
Shenzhen Tendfo Medical Technology Co Ltd
Shenzhen Tengfu Medical Technology Co Ltd
Suzhou Tianhong Shengjie Medical Co Ltd
TransMed7 LLC
University of Arizona
University of California Los Angeles
University of Michigan
University of Toledo
University of Wolverhampton
VeinWay Ltd
Veniti Inc
VueKlar Cardiovascular Ltd (Inactive)
Wallaby Medical Inc
WaveClear Inc
Zylox-Tonbridge Medical Technology Co Ltd
Table of Contents
Table
Figures
Frequently asked questions
-
Which was the leading segment in the clot management devices pipeline market as of February 2024?
Thrombectomy Systems (Catheters) was the leading segment in the clot management devices pipeline market as of February 2024.
-
Which territory had the highest number of products in the clot management devices pipeline market as of February 2024?
As of February 2024, the US had the highest number of products in the clot management devices pipeline market.
-
Which was the most followed regulatory pathway in the clot management devices pipeline market as of February 2024?
As of February 2024, 510(k) was the most followed pathway for pipeline products in the clot management devices market.
-
Which are the leading companies operating in the clot management devices pipeline market?
The key companies in the clot management devices pipeline market are AorticLab Sarl, APT Medical Inc, Boston Scientific Corp, Caeli Vascular LLC, Capture Vascular Inc, Embolx, Inc., and Inari Medical Inc. among others.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more Clot Management Devices reports