Cell and Gene Therapies in CNS Disorders – Disease Overview, Treatment Options and Future Market Assessment
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
Cell and Gene Therapies in CNS Disorders Market Report Overview
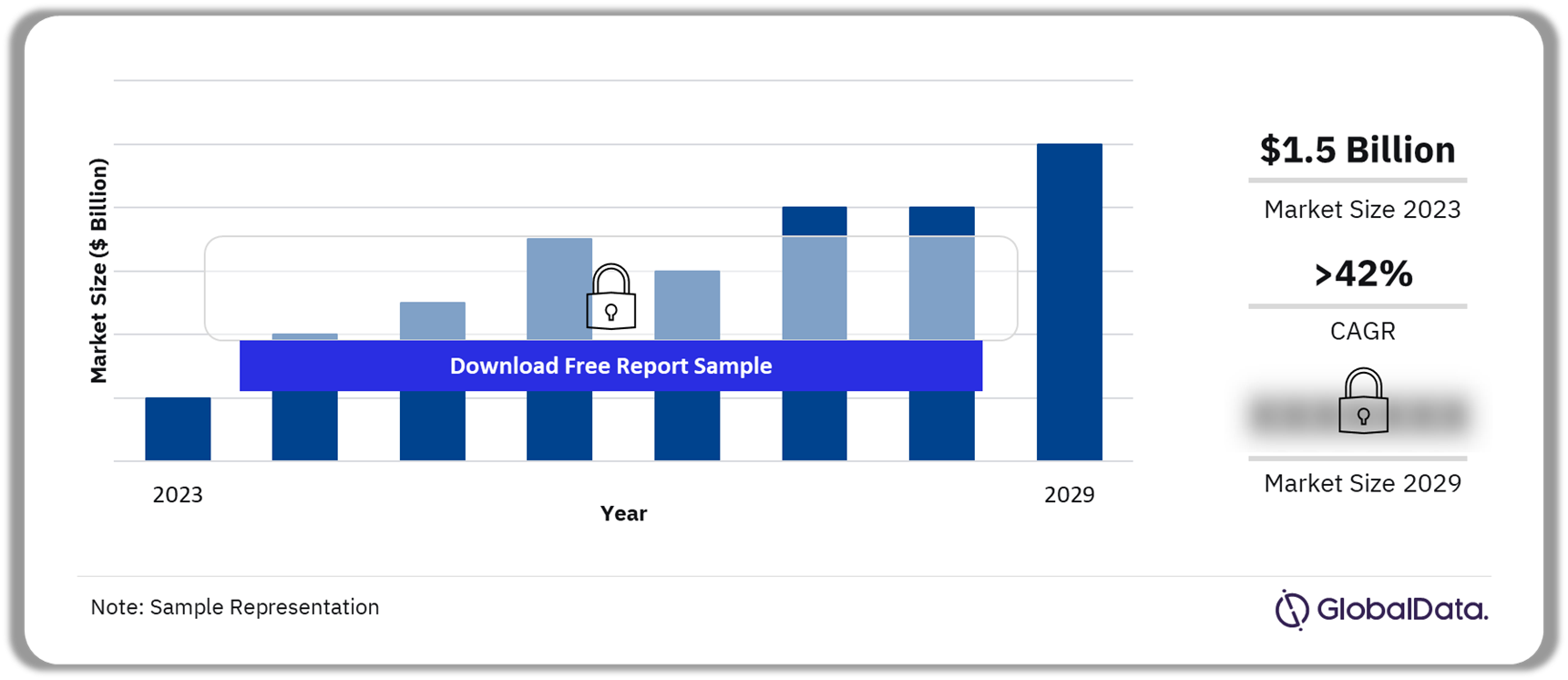
The cell and gene therapies (CGT) sales in central nervous system (CNS) disorders was $1.5 billion in 2023. It will experience significant growth, achieving a CAGR of more than 42% during the forecast period. Gene therapies will dominate the CGT market in CNS disorders because of strong players such as Sarepta Therapeutics and Novartis with marketed products Elevidys and Zolgensma, respectively.
Cell and Gene Therapies in CNS Disorders Market Outlook 2023-2029 (Billion)
Buy the Full Report for More Insights on the CNS Disorders Market
The CGT in CNS disorders market research report will aid clients in gaining insight into the competitive landscape of leading CGT agents in CNS disorders including launch date projections, key clinical trial analysis, analyst consensus forecasts, likelihood of approval analysis, and commentary on current and future players. The report also includes an outlook of pricing and reimbursement assessment. Additionally, the report includes regulations of CGTs in 8MM, FDA guidance, and future market assessment.
| Key Companies | · Novartis
· PTC Therapeutics · Kyowa Kirin · Bluebird bio |
| Enquire & Decide | Discover the perfect solution for your business needs. Enquire now and let us help you make an informed decision before making a purchase. |
Cell and Gene Therapies (CGT) in CNS Disorders Market Dynamics
Various factors could impact the CGT in CNS Disorders such as multigene disorders are difficult to target with CGTs; early disease detection and CGT intervention; administration of CGTs, and others
Early disease detection and CGT intervention: To increase the likelihood of CGT success, it must be delivered during the early stages of the disorder, during the prodromal stages, or when patients are pre-symptomatic. KOLs noted that particularly for rare diseases, there is a delay in patient referral to specialist treatment. Owing to this, greater initiatives are required to help with early screening.
Buy the Full Report for More Insights on the CGT in CNS Disorders Market Dynamics
Cell and Gene Therapies (CGT) in CNS Disorders – Current Treatment Options
There are only three cell therapies that have received conditional approval for CNS disorders. These therapies target disorders with a high prevalence and are not monogenic disorders. KOLs agreed that research into the application of gene therapies in CNS disorders is more advanced than cell therapies. Although these therapies have received conditional approval, KOLs noted that access to these cell therapies was limited and often restricted to a single hospital. As such, very few patients have access to such treatments.
Patient assistance programs specifically focusing on treatment journey navigation are offered across all approved therapies as the primary focus of commercial activities. This is because of the complicated route to diagnosis, treatment, and access pathways associated with CGTs.
Cell and Gene Therapies (CGT) in CNS Disorders – Future Market Assessment
- Gene therapies are in development to target rare genetic CNS disorders including Duchenne muscular dystrophy, Friedreich ataxia, Rett syndrome, and Batten disease.
- Duchenne muscular dystrophy has two Phase III assets in development, while amyotrophic lateral sclerosis has one Phase III product and one Phase II/III in development. In contrast, there are no Phase III CGT assets for Parkinson’s disease, Alzheimer’s disease, or multiple sclerosis. KOLs suspected that these top five disorders have a highly prevalent patient population and/or a lack of disease-modifying treatments available, making them key indications for the development of CGT therapies.
Buy the Full Report for More Analysis on the Current Treatment Options and Future Market Assessment of CGT in CNS Disorders
Cell and Gene Therapies (CGT) in CNS Disorders – Competitive Landscape
The key companies in the CGT in CNS disorders market are Novartis, PTC Therapeutics, Kyowa Kirin, and Bluebird bio among others.
Novartis’s Zolgensma for the treatment of spinal muscular atrophy (SMA) was the first CGT asset to receive traditional approval in the CNS market in 2019. Over the past five years, there has been an increase in the number of traditionally approved CGTs for genetic disorders.
Key CGT Companies for CNS Disorders
Buy the Full Report for More Insights on the Key CGT in CNS Disorders Players
Scope
Overview of cell and gene therapies (CGT) in CNS Disorders. The report focuses on the marketed CGT assets addressing CNS disorders as well as the top five CNS indications with the most CGT pipeline assets. Clients will gain insight into the competitive landscape of leading CGT agents in CNS disorders including launch date projections, analyst consensus forecasts, likelihood of approval analysis, and commentary on current and future players. The report also includes outlook from three key opinion leaders in the 5EU, US, and Japan, and analyses key challenges and opportunities in the application of CGTs in CNS disorders. Additionally, the report includes commentary on the regulatory landscape of CGTs and on the reimbursement environment.
Key Highlights
Report deliverable includes a PowerPoint report.
Reasons to Buy
• Develop and design your in-licensing and out-licensing strategies through a review of pipeline products and technologies, and by identifying the companies with the most robust pipeline.
• Develop business strategies by understanding the trends shaping and driving the CGT in CNS market.
• Drive revenues by understanding the key trends, innovative products and technologies, market segments, and companies likely to impact the CNS disorder targeting CGT therapeutics market in the future.
• Formulate effective sales and marketing strategies by understanding the competitive landscape and by analyzing the performance of various competitors.
• Identify emerging players with potentially strong product portfolios and create effective counterstrategies to gain a competitive advantage.
• Organize your sales and marketing efforts by identifying the market categories and segments that present maximum opportunities for consolidations, investments, and strategic partnerships.
Sarepta Therapeutics Inc
PTC Therapeutics
Kyowa Kirin Co Ltd
bluebird bio Inc
Corestemchemon Inc
Biostar Stem Cell Research Institute
Nipro Corp
Pfizer Inc
Capricor Therapeutics Inc
Rapa Therapeutics LLC
BrainStorm Cell Therapeutics Inc
Table of Contents
Frequently asked questions
-
What were the CGT sales in the CNS disorders in 2023?
The CGT sales for CNS disorders were $1.5 billion in 2023.
-
Which was the first CGT asset to receive traditional approval in the CNS market in 2019?
Novartis’s Zolgensma for the treatment of spinal muscular atrophy (SMA) was the first CGT asset to receive traditional approval in the CNS market in 2019.
-
Which are the factors impacting the CGT in CNS disorders?
A few of the factors impacting CGT in CNS disorders are difficult to target with CGTs; early disease detection and CGT intervention; administration of CGTs, and others.
-
Which are the key players in the CGT in CNS disorders market?
The key players in the CGT in CNS disorders market are Novartis, PTC Therapeutics, Kyowa Kirin, and Bluebird bio among others.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more Central Nervous System reports