Dental Bone Graft Substitutes and Regenerative Materials Pipeline Report including Stages of Development, Segments, Region and Countries, Regulatory Path and Key Companies, 2022 Update
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
Dental bone grafts and substitutes are used to augment or fill a defect created by surgery, trauma, disease, or developmental deficiency. Bone grafts and regenerative materials regenerate the natural bone and dental tissue.
The dental bone graft substitutes & regenerative materials pipeline market research report provides comprehensive information about the dental bone graft substitutes & regenerative materials pipeline products with a comparative analysis of the products at various stages of development and information about the clinical trials which are in progress. Moreover, the report provides information about various pipeline products and their estimated approval dates.
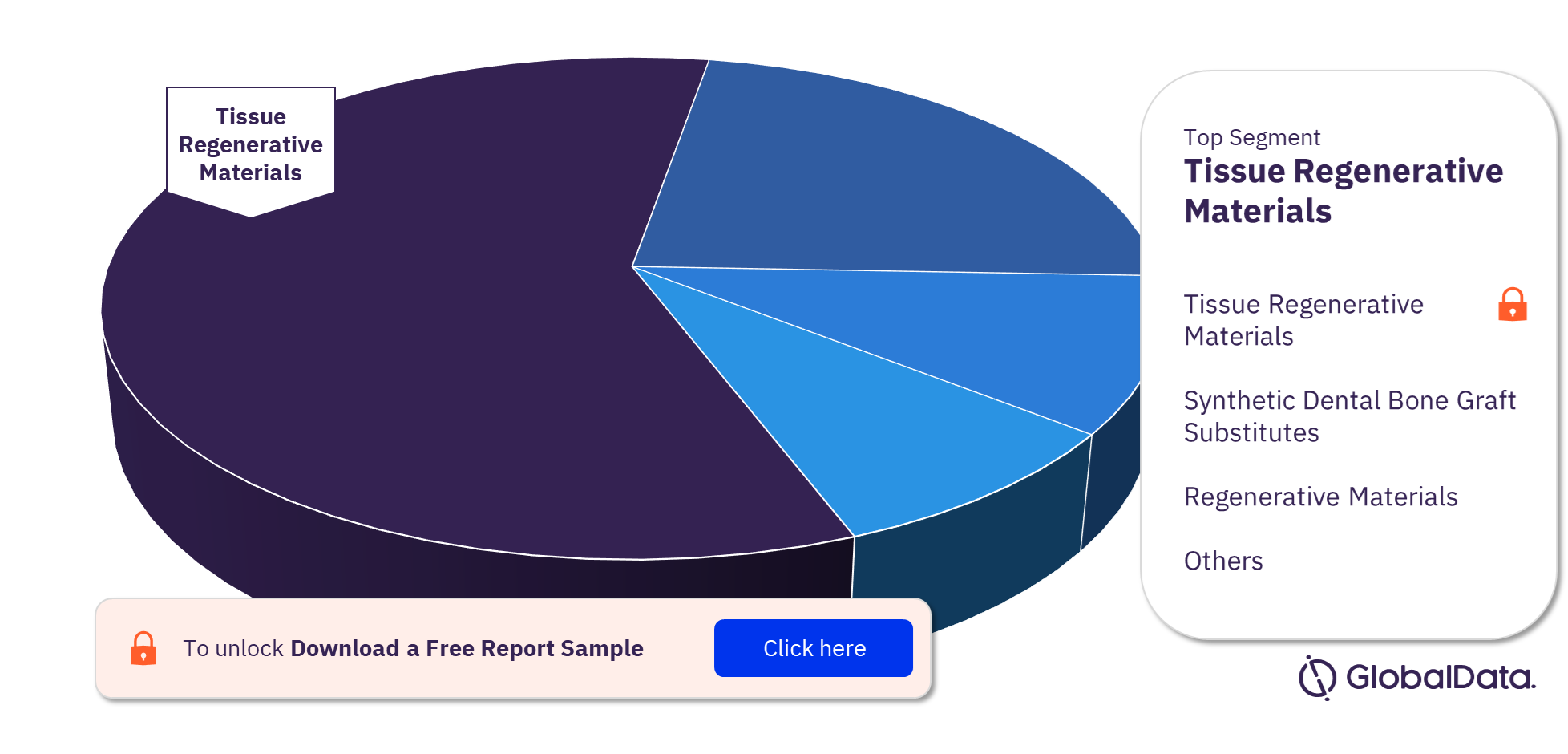
Dental Bone Graft Substitutes & Regenerative Materials Pipeline Products Market Segments
The key segments in the dental bone graft substitutes & regenerative materials pipeline products market are tissue regenerative materials, synthetic dental bone graft substitutes, dental bone graft substitutes & dental materials, regenerative materials, and dental bone grafts. Tissue regenerative materials has the highest number of pipeline products in the dental bone graft substitutes & regenerative materials pipeline products market.
Regenerative Materials: Regenerative materials are biomaterials that facilitate the regeneration of lost periodontal hard and soft tissues. Regenerative materials include tissue regenerative materials and growth factors.
Dental Bone Grafts: Dental bone graft is used to augment or fill a defect created by surgery, trauma, disease, and developmental deficiency. These products are available in the form of blocks, gels, pastes, particulates, putty, and strips.
Dental Bone Graft Substitutes & Regenerative Materials Pipeline Products Market Analysis by Segments, 2022 (%)
For more segment insights into the dental bone graft substitutes & regenerative materials pipeline products market, download a free report sample
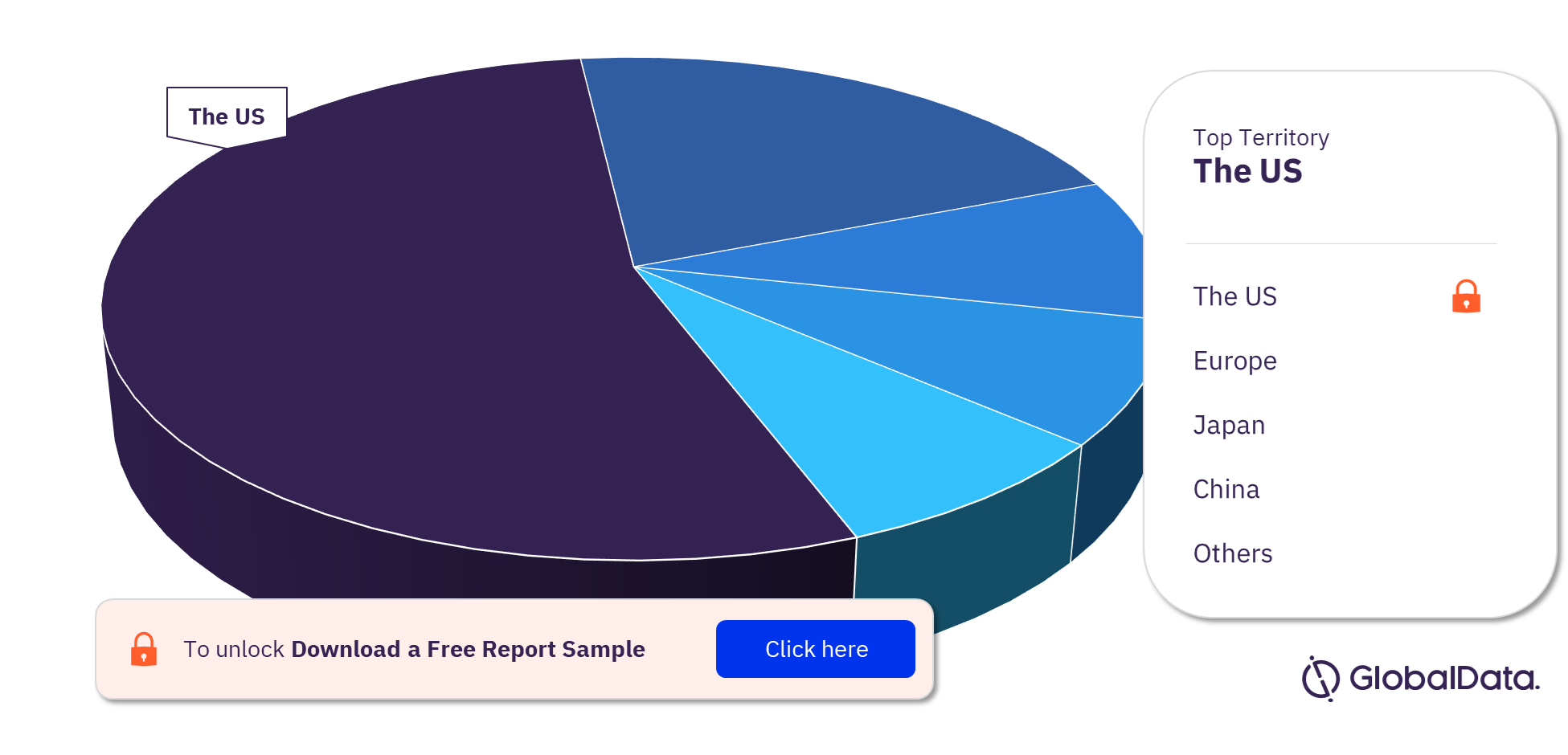
Dental Bone Graft Substitutes & Regenerative Materials Pipeline Products Market Segmentation by Territories
Some of the key territories with products in the pipeline are the US, Europe, Japan, China, New Zealand, Taiwan, the UK, India, Australia, and Canada. The US is the leading territory in the dental bone graft substitutes & regenerative materials pipeline products market.
Dental Bone Graft Substitutes & Regenerative Materials Pipeline Products Market Analysis by Territories, 2022 (%)
For more territory insights into the dental bone graft substitutes & regenerative materials pipeline products market, download a free report sample
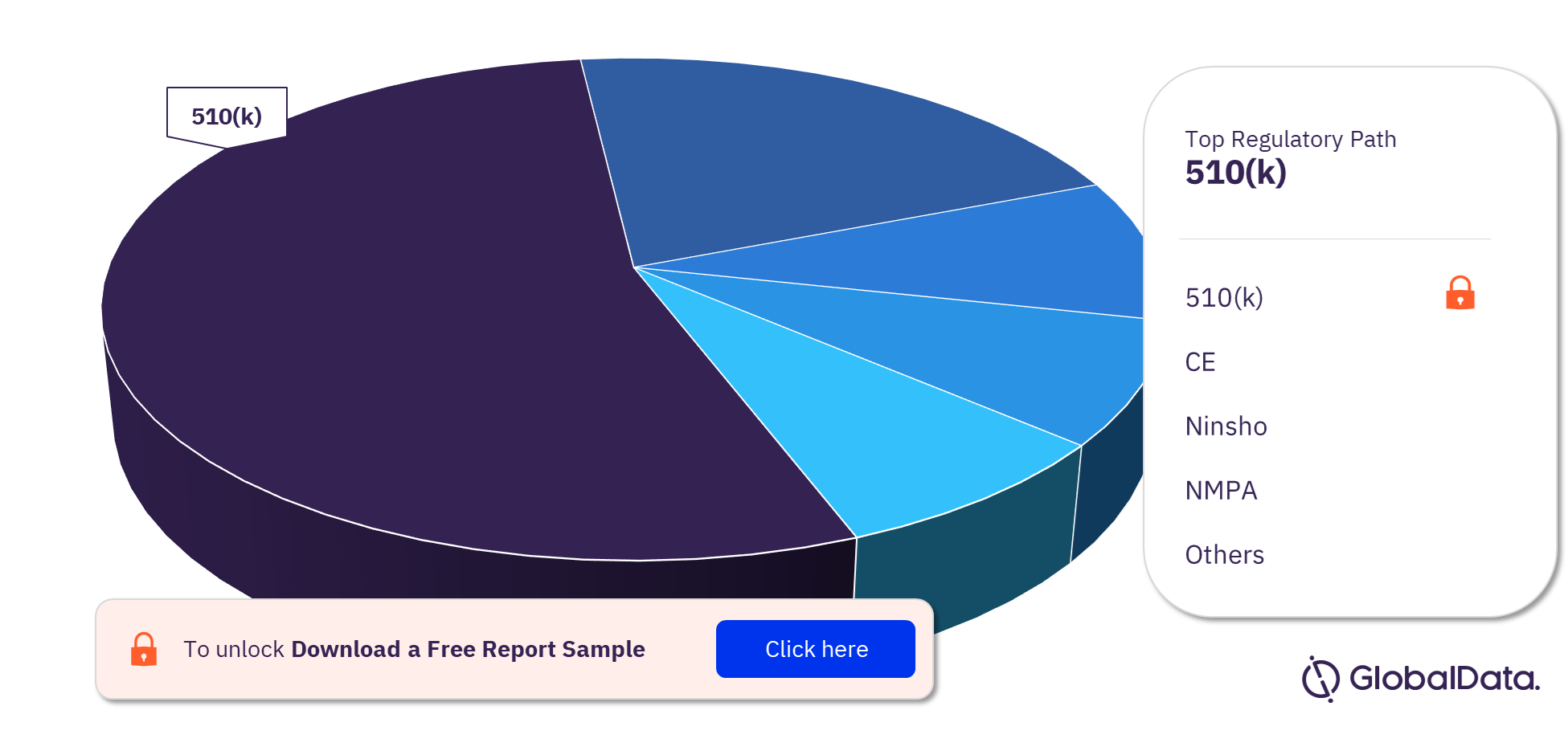
Dental Bone Graft Substitutes & Regenerative Materials Pipeline Products Market Segmentation by Regulatory Paths
The dental bone graft substitutes & regenerative materials pipeline report provides detailed insights into the pipeline products by a regulatory path including 510(k), CE, Ninsho, NMPA, PMA, TGA, UKCA, ICAC, MDL, and BOPA. Most of the products follow the 510(k) pathway to enter the market.
Dental Bone Graft Substitutes & Regenerative Materials Pipeline Products Market Analysis by Regulatory Paths, 2022 (%)
For more regulatory path insights into the dental bone graft substitutes & regenerative materials pipeline products market, download a free report sample
Dental Bone Graft Substitutes & Regenerative Materials Pipeline Products Market - Competitive Landscape
Some of the leading companies in the dental bone graft substitutes & regenerative materials pipeline products market are 3-D Matrix Ltd, Advanced Biotech Products Pvt Ltd, Cayman Chemical Co, CellSeed Inc, Colorado Therapeutics, LLC, Fujifilm Corp, Histocell SL, Indian Institute of Technology Bombay, Induce Biologics Inc., and Ivory Graft Ltd.
Dental Bone Graft Substitutes & Regenerative Materials Pipeline Products Market Report Overview
| Key Territories | The US, Europe, Japan, China, New Zealand, Taiwan, the UK, India, Australia, and Canada |
| Key Regulatory Paths | 510(k), CE, Ninsho, NMPA, PMA, TGA, UKCA, ICAC, MDL, and BOPA |
| Key Segments | Tissue Regenerative Materials, Synthetic Dental Bone Graft Substitutes, Dental Bone Graft Substitutes & Dental Materials, Regenerative Materials, and Dental Bone Grafts |
| Leading Companies | 3-D Matrix Ltd, Advanced Biotech Products Pvt Ltd, Cayman Chemical Co, CellSeed Inc, Colorado Therapeutics, LLC, Fujifilm Corp, Histocell SL, Indian Institute of Technology Bombay, Induce Biologics Inc., and Ivory Graft Ltd |
Segments Covered in the Report
Dental Bone Graft Substitutes & Regenerative Materials Pipeline Products Market Segments Outlook
- Tissue Regenerative Materials
- Synthetic Dental Bone Graft Substitutes
- Dental Bone Graft Substitutes & Dental Materials
- Regenerative Materials
- Dental Bone Grafts
Dental Bone Graft Substitutes & Regenerative Materials Pipeline Products Market Territories Outlook
- The US
- Europe
- Japan
- China
- New Zealand
- Taiwan
- the UK
- India
- Australia
- Canada
Dental Bone Graft Substitutes & Regenerative Materials Pipeline Products Market Regulatory Paths Outlook
- 510(k)
- CE
- Ninsho
- NMPA
- PMA
- TGA
- UKCA
- ICAC
- MDL
- BOPA
Scope
- Extensive coverage of the dental bone graft substitutes & regenerative materials under development
- The report reviews details of major pipeline products which includes, product description, licensing and collaboration details, and other developmental activities
- The report reviews the major players involved in the development of dental bone graft substitutes & regenerative materials and lists all their pipeline projects
- The coverage of pipeline products is based on various stages of development ranging from early development to the approved/issued stage
- The report provides key clinical trial data of ongoing trials specific to pipeline products
- Recent developments in the segment/industry
Reasons to Buy
The report enables you to:
- Formulate significant competitor information, analysis, and insights to improve R&D strategies
- Identify emerging players with potentially strong product portfolios and create effective counter strategies to gain a competitive advantage
- Identify and understand important and diverse types of dental bone graft substitutes & regenerative materials under development
- Develop market-entry and market expansion strategies
- Plan mergers and acquisitions effectively by identifying major players with the most promising pipeline
- In-depth analysis of the product’s current stage of development, territory, and estimated launch date
Advanced Biotech Products Pvt Ltd
Cayman Chemical Co
CellSeed Inc
Colorado Therapeutics, LLC
credentis AG
curasan AG
Fujifilm Corp
Histocell SL
Indian Institute of Technology Bombay
Induce Biologics Inc.
Ivory Graft Ltd
Malla Reddy Institute of Medical Sciences
Maxigen Biotech Inc
Medtronic Plc
Nobel Biocare Services AG
Novatissue SAS
Orthocell Ltd
Orthogen LLC
Orthorebirth Co Ltd
Orthosera GmbH
PolyPid Ltd
Polytechnic University of Catalonia
RevBio Inc
Sree Chitra Tirunal Institute for Medical Sciences & Technology
Straumann Holding AG
SurgaColl Technologies Ltd
SweetBio, Inc.
Tetratherix Pty Ltd
Tigran Technologies AB
University of Bielefeld
University of Connecticut Health Center
University of Maryland
ZetaGen Therapeutics Inc
Table of Contents
Table
Figures
Frequently asked questions
-
Which are the key territories in the dental bone graft substitutes & regenerative materials pipeline products market?
Some of the key territories in the dental bone graft substitutes & regenerative materials pipeline products market are the US, Europe, Japan, China, New Zealand, Taiwan, the UK, India, Australia, and Canada.
-
What are the key regulatory paths in the dental bone graft substitutes & regenerative materials pipeline products market?
The key regulatory paths in the dental bone graft substitutes & regenerative materials pipeline products market are 510(k), CE, Ninsho, NMPA, PMA, TGA, UKCA, ICAC, MDL, and BOPA.
-
What are the key segments in the dental bone graft substitutes & regenerative materials pipeline products market?
The key segments in the dental bone graft substitutes & regenerative materials pipeline products market are tissue regenerative materials, synthetic dental bone graft substitutes, dental bone graft substitutes & dental materials, regenerative materials, and dental bone grafts.
-
Which are the leading companies in the dental bone graft substitutes & regenerative materials pipeline products market?
Some of the leading companies in the dental bone graft substitutes & regenerative materials pipeline products market are 3-D Matrix Ltd, Advanced Biotech Products Pvt Ltd, Cayman Chemical Co, CellSeed Inc, Colorado Therapeutics, LLC, Fujifilm Corp, Histocell SL, Indian Institute of Technology Bombay, Induce Biologics Inc., and Ivory Graft Ltd.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more Dental Devices reports