Knee Reconstruction Pipeline by Stages of Development, Segments, Region, Regulatory Path and Key Companies
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
Knee Reconstruction Pipeline Market Report Overview
Knee Reconstruction is a surgical procedure involving the replacement of damaged or diseased parts of the knee joint. This category includes devices (implants) used in primary, partial and revision knee replacement procedures.
The knee reconstruction pipeline market research report provides comprehensive information about the Knee reconstruction pipeline products with a comparative analysis of the products at various stages of development and information about the clinical trials which are in progress.
| Key Segments | · Primary Knee Replacement
· Revision Knee Replacement · Partial Knee Replacement · Primary Cementless Knee Replacement · Complete Hinged Revision Knee Replacement · Patello-Femoral Partial Knee Replacement · Primary Cemented Knee Replacement |
| Key Territories | · The US
· China · Europe · Australia · New Zealand · Singapore · Others |
| Key Regulatory Paths | · 510(k)
· NMPA · CE · PMA · TGA · Shonin · Others |
| Leading Companies | · 4WEB Medical Inc
· Active Implants LLC · CEA-Leti · ConforMIS Inc · Delta Orthopedics LLC · Fuse Medical Inc · Others |
| Enquire & Decide | Discover the perfect solution for your business needs. Enquire now and let us help you make an informed decision before making a purchase. |
Knee Reconstruction Pipeline Market by Segments
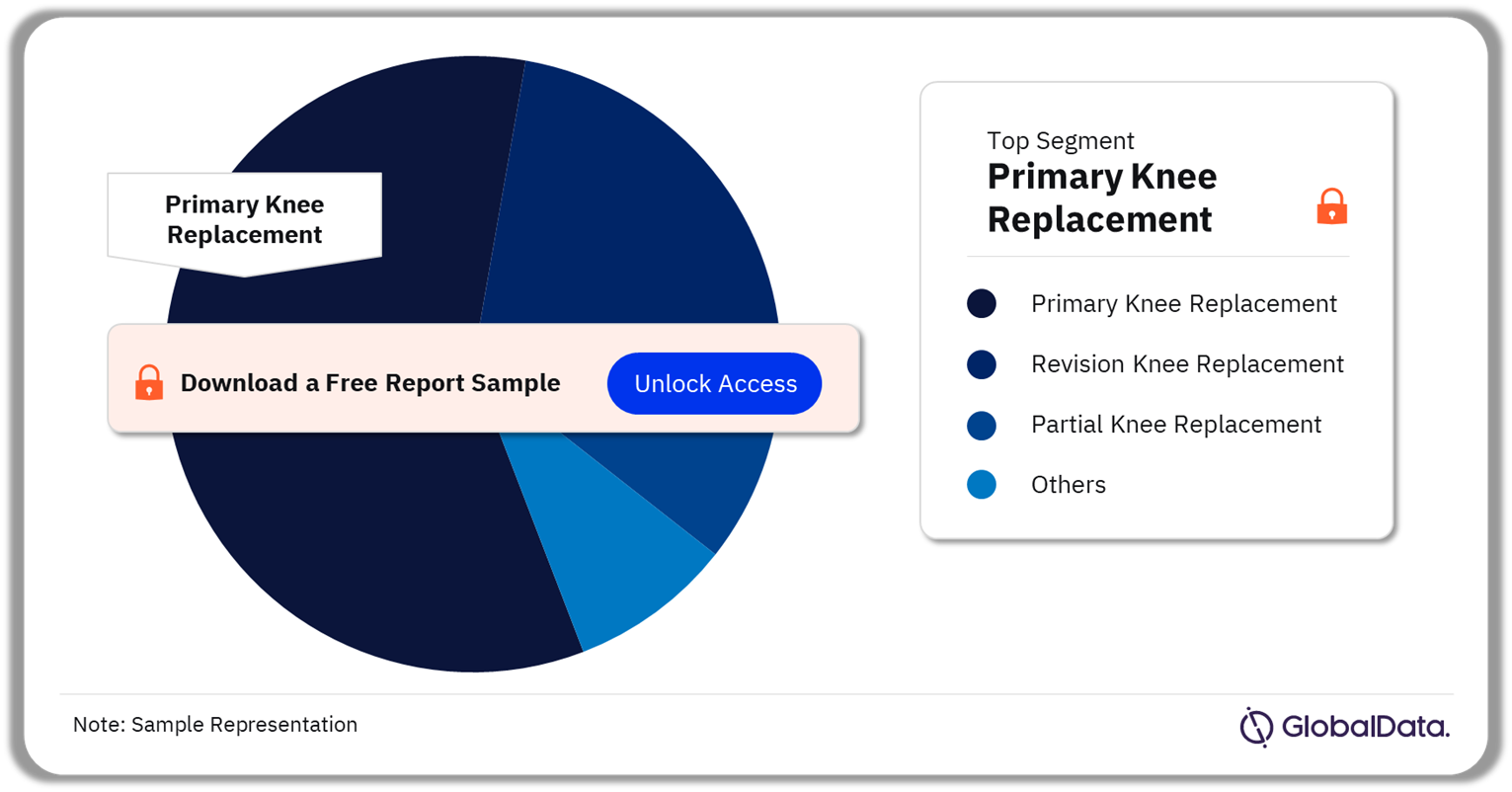
The key segments in the knee reconstruction market are primary knee replacement, revision knee replacement, partial knee replacement, primary cementless knee replacement, complete hinged revision knee replacement, patello-femoral partial knee replacement, and primary cemented knee replacement. As of July 2023, the primary knee replacement segment had the highest number of pipeline products. Also known as total knee arthroplasty or total knee replacement, the typical primary knee replacement implant set consists of four components: a metal femoral component, a metal tibial component, a polyethylene inserts or spacer, and a patellar component. One unit is one implant set.
Knee Reconstruction Pipeline Market Analysis by Segments, 2023 (%)
For more segment insights into the Knee reconstruction pipeline market, download a free report sample
Knee Reconstruction Pipeline Market Segmentation by Territories
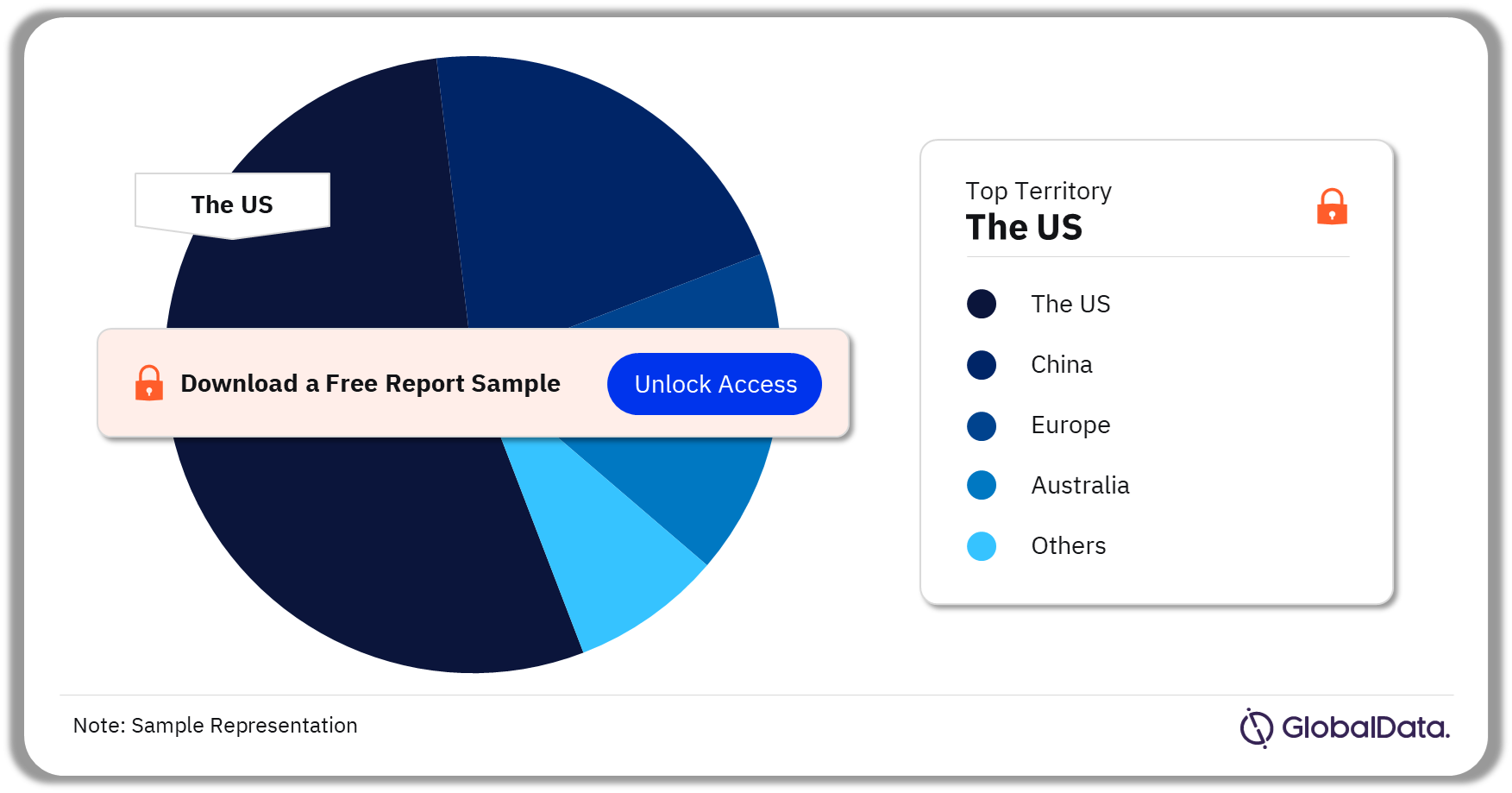
Some of the key territories in the Knee reconstruction market are the US, China, Europe, Australia, New Zealand, Singapore, and South Korea among others. As of July 2023, the United States had the highest number of pipeline products.
Knee Reconstruction Pipeline Market Analysis by Territories, 2023 (%)
For more territory insights into the Knee reconstruction pipeline market, download a free report sample
Knee Reconstruction Pipeline Market Segmentation by Regulatory Paths
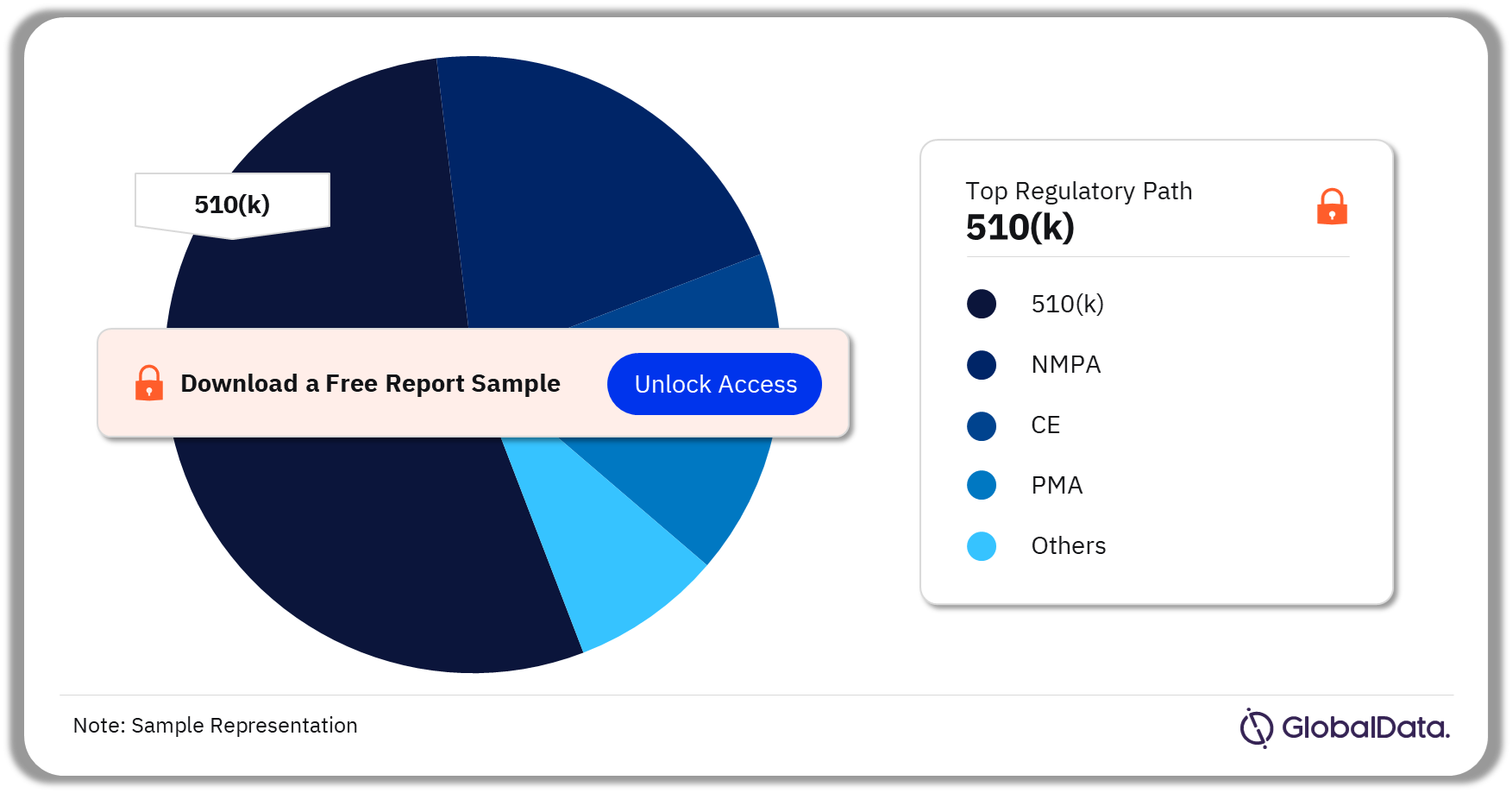
Some of the key regulatory paths in the Knee reconstruction pipeline market are 510(k), NMPA, CE, PMA, TGA, and Shonin, among others. As of July 2023, 510(k) was the most followed pathway for pipeline products.
Knee Reconstruction Pipeline Market Analysis by Regulatory Paths, 2023 (%)
For more regulatory path insights into the knee reconstruction pipeline market, download a free report sample
Knee reconstruction Pipeline Market – Competitive Landscape
Some of the key companies in the Knee reconstruction pipeline market are 4WEB Medical Inc, Active Implants LLC, CEA-Leti, ConforMIS Inc, Delta Orthopedics LLC, and Fuse Medical Inc among others.
4WEB Medical Inc: 4WEB Medical Inc (4WEB) is an orthopedic implant device company that designs 3D printed titanium implants. The company also designs and develops implants for trauma, tumor, and patient-specific applications. Its product portfolio includes anterior, posterior, cervical, and lateral spine truss systems, as well as the osteotomy truss system and implant custom services for extremity, tumor and trauma procedures. It utilizes 3D printing technology to develop 4WEB Medical’s proprietary truss implant platform. 4WEB is headquartered in Frisco, Texas, the US.
Active Implants LLC: Active Implants LLC (Active Implants), formerly Active Implants Corp, is a medical equipment provider that develops orthopedic equipment. The company develops and commercializes polymer orthopedic implant solutions which include tribofit hip systems, tribofit instrumentation equipment, NuSurface meniscus implant and others. It develops and markets low wear and bone spurting orthopedic implant solutions that are made of polyurethane materials. Active Implants’ joint replacement products are manufactured using poly-carbonate urethane materials. The company operates in the Netherlands, Israel, and others. Active Implants is headquartered in Memphis, Tennessee, the US.
Leading Knee Reconstruction Companies, 2023
For more company insights into the Knee reconstruction pipeline market, download a free report sample
Segments Covered in the Report
Knee Reconstruction Pipeline Market Segments Outlook
- Primary Knee Replacement
- Revision Knee Replacement
- Partial Knee Replacement
- Primary Cementless Knee Replacement
- Complete Hinged Revision Knee Replacement
- Patello-Femoral Partial Knee Replacement
- Primary Cemented Knee Replacement
Knee Reconstruction Pipeline Market Territories Outlook
- The US
- China
- Europe
- Australia
- New Zealand
- Singapore
- Others
Knee Reconstruction Pipeline Market Regulatory Paths Outlook
- 510(k)
- NMPA
- CE
- PMA
- TGA
- Shonin
- Others
Scope
This report provides:
- Extensive coverage of the Knee reconstruction under development.
- Details of major pipeline products which includes product description, licensing and collaboration details, and other developmental activities.
- Reviews of the major players involved in the development of Knee reconstruction and lists of all their pipeline projects.
- Coverage of pipeline products based on various stages of development ranging from Early Development to Approved/Issued stage.
- Key clinical trial data of ongoing trials specific to pipeline products.
- Recent developments in the segment/industry.
Reasons to Buy
The report enables you to:
- Formulate significant competitor information, analysis, and insights to improve R&D strategies.
- Identify emerging players with potentially strong product portfolios and create effective counter strategies to gain a competitive advantage.
- Identify and understand important and diverse types of Knee reconstruction under development.
- Develop market-entry and market-expansion strategies.
- Plan mergers and acquisitions effectively by identifying major players with the most promising pipeline.
- In-depth analysis of the product’s current stage of development, territory, and estimated launch date
Active Implants LLC
Artimplant AB (Inactive)
Aspect Biosystems Ltd
Bactiguard Holding AB
Binghamton University
BioPoly LLC
Bonovo Orthopedics Inc
CEA-Leti
CeramTec GmbH
Clemson University
ConforMIS Inc
Corin Group Ltd
CytexOrtho
Delta Orthopedics LLC
DiFusion Technologies Inc
Ecole de technologie superieure
Elite Surgical Supplies (Pty) Ltd
Episurf Medical AB
Exactech Inc
EZ ORTHOPEDICS ISRAEL
ForCast Orthopedics Inc
Fuse Medical Inc
Ganymed Robotics
Genovel Orthopedics Inc
Georgia Institute of Technology
Hospital for Special Surgery
Indian Institute of Technology Bombay
Invibio Knee Ltd
Kinamed Inc
Materialise NV
Materials Mines Paris Tech Center
Medacta Group SA
MicroPort Orthopedics Inc
MicroPort Scientific Corp
Monogram Orthopaedics Inc
Nano Interface Technology LLC (Inactive)
NovoPedics, Inc.
Ohio State University
Okani Medical Technology
OrthoGrid Systems
Orthonika Ltd
OrthoSensor Inc
Resultados y Calidad del Sistema Sanitario Publico de Andalucia
RIH Orthopaedic Foundation Inc
Sanatmetal Ltd.
Sensortech Corp
SINTX Technologies Inc
Smith & Nephew Inc
Smith & Nephew Plc
Stellenbosch University
Stevens Institute of Technology
University of Bath
University of Florida
University of South Carolina
Table of Contents
Table
Figures
Frequently asked questions
-
Which was the leading segment in the knee reconstruction pipeline market?
Primary knee replacement was the leading segment in the knee reconstruction pipeline market.
-
Which territory has the highest number of pipeline products in the knee reconstruction pipeline market?
As of July 2023, the US had the highest number of pipeline products in the knee reconstruction pipeline market.
-
Which was the most followed regulatory pathway in the knee reconstruction pipeline market?
As of July 2023, 510(k) was the most followed regulatory pathway in the knee reconstruction pipeline market.
-
Who are the major players operating in the knee reconstruction pipeline market?
Some of the key companies in the Knee reconstruction pipeline market are 4WEB Medical Inc, Active Implants LLC, CEA-Leti, ConforMIS Inc, Delta Orthopedics LLC, and Fuse Medical Inc.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more Knee Reconstruction reports