Pedicle Screw-Based Dynamic Stabilization Systems Pipeline by Stages of Development, Segments, Region, Regulatory Path and Key Companies
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
Pedicle Screw-Based Dynamic Stabilization Systems Pipeline Market Report Overview
Pedicle Screw-Based Dynamic stabilization systems include two components, one for attachment to a superior vertebra of a spinal motion segment and the other for the attachment to an inferior vertebra of a spinal motion segment. Pedicle screw-based dynamic stabilization system restores stability after decompression. The device includes screws, spacer, and cords.
The Pedicle screw-based dynamic stabilization systems pipeline market research report provides comprehensive information about the Pedicle screw-based dynamic stabilization systems pipeline products with a comparative analysis of the products at various stages of development and information about the clinical trials that are in progress.
| Key Territories | · The US
· Europe · The UK · Canada · China |
| Key Regulatory Paths | · 510(k)
· PMA · CE · MDL · NMPA · UKCA |
| Leading Companies | · ARO Medical, LLC
· Colorado State University · Curtin University · Jemo Spine, LLC. · Medtronic Plc |
| Enquire & Decide | Discover the perfect solution for your business needs. Enquire now and let us help you make an informed decision before making a purchase. |
Pedicle Screw-Based Dynamic Stabilization Systems Pipeline Market Segmentation by Territories
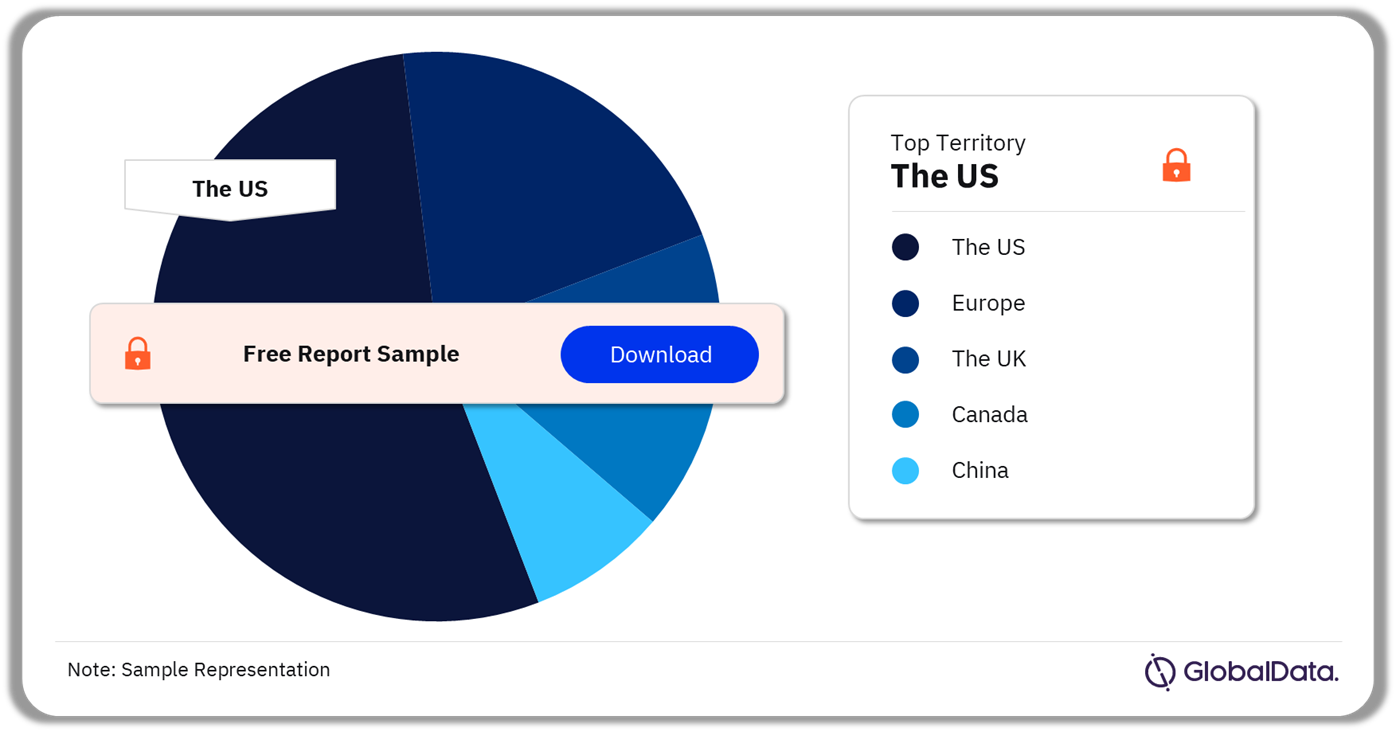
Some of the key territories in the pedicle screw-based dynamic stabilization systems market are the US, Europe, the UK, Canada, and China. As of November 2023, the US has the highest number of pipeline products.
Pedicle Screw-Based Dynamic Stabilization Systems Pipeline Market Analysis by Territories, 2023 (%)
Buy the Full Report for More Territory Insights into the Pedicle Screw-Based Dynamic Stabilization Systems Pipeline Market
Pedicle Screw-Based Dynamic Stabilization Systems Pipeline Market Segmentation by Regulatory Paths
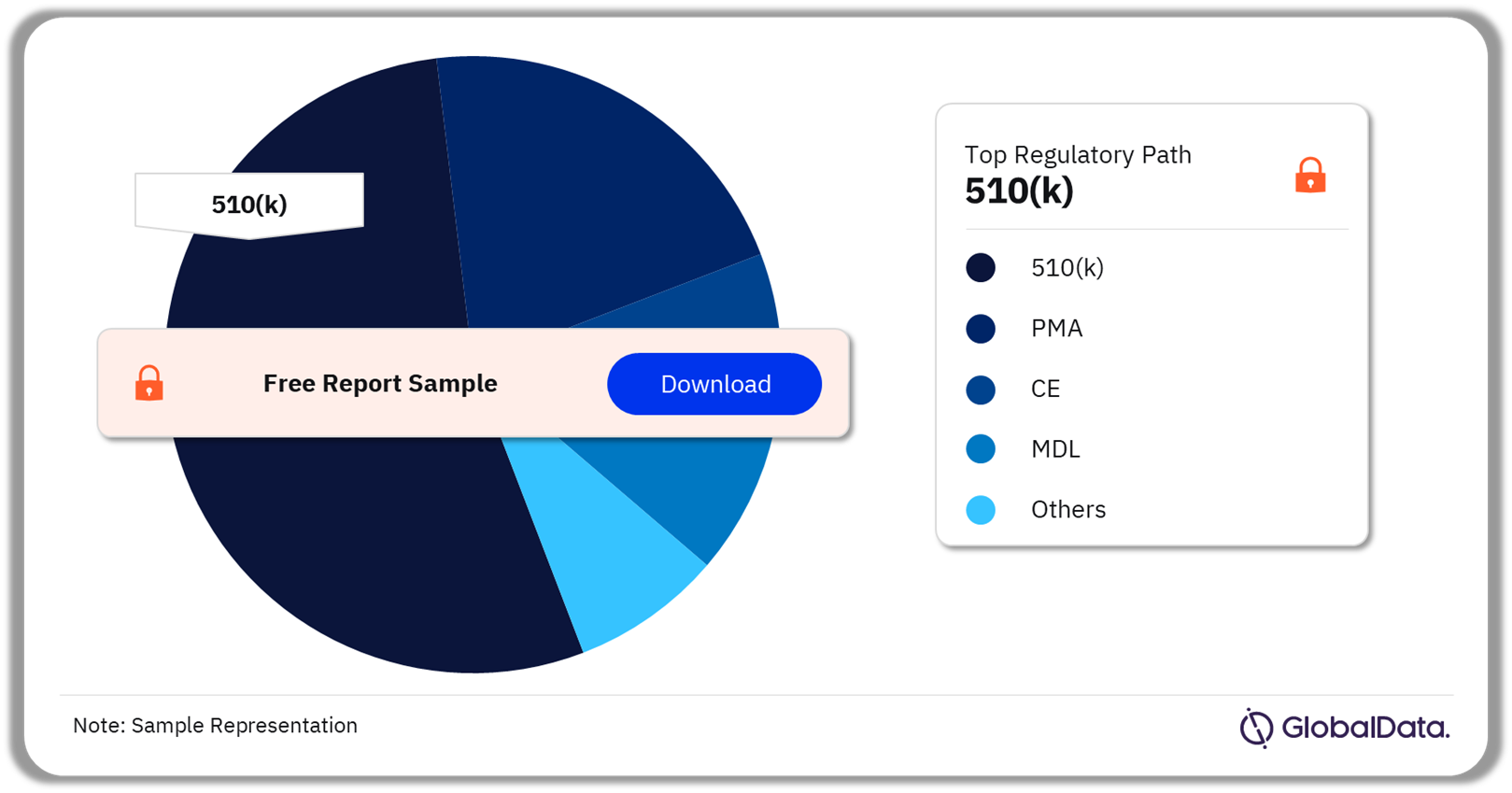
Some of the key regulatory paths in the pedicle screw-based dynamic stabilization systems pipeline market are 510(k), PMA, CE, MDL, NMPA, and UKCA. As of November 2023, 510(k) is the most followed pathway for pipeline products in the market.
Pedicle Screw-Based Dynamic Stabilization Systems Pipeline Market Analysis by Regulatory Paths, 2023 (%)
Buy the Full Report for More Regulatory Path Insights into the Pedicle Screw-Based Dynamic Stabilization Systems Pipeline Market
Pedicle Screw-Based Dynamic Stabilization Systems Pipeline Market – Competitive Landscape
Some of the key companies in the pedicle screw-based dynamic stabilization systems pipeline market are ARO Medical, LLC, Colorado State University, Curtin University, Jemo Spine, LLC, and Medtronic Plc among others.
Jemo Spine, LLC: Jemo Spine, LLC. (Jemo Spine) is a medical device company that designs, develops, manufactures, and markets orthopedic products for the spine market. The company’s products include a spinal fusion system, anterior cervical plate, and interbody spacer, interspinous decompression system, dynamic decompression and stabilization system, flexible rod system, and synthetic bone substitutes, among others.
Pedicle Screw-Based Dynamic Stabilization Systems Pipeline Market Analysis by Companies, 2023
Buy the Full Report for More Company Insights into the Pedicle Screw-Based Dynamic Stabilization Systems Pipeline Market
Segments Covered in the Report
Pedicle Screw-Based Dynamic Stabilization Systems Pipeline Market Territories Outlook
- The US
- Europe
- The UK
- Canada
- China
Pedicle Screw-Based Dynamic Stabilization Systems Pipeline Market Regulatory Paths Outlook
- 510(k)
- PMA
- CE
- MDL
- NMPA
- UKCA
Scope
This report provides:
- Extensive coverage of the pedicle screw-based dynamic stabilization systems under development.
- Details of major pipeline products which include product description, licensing and collaboration details and other developmental activities.
- Reviews of major players involved in the development of pedicle screw-based dynamic stabilization systems and lists all their pipeline projects.
- Coverage of pipeline products based on various stages of development ranging from early development to approved/issued stage.
- Provides key clinical trial data of ongoing clinical trials specific to pipeline products.
- Recent developments in the segment/industry.
Reasons to Buy
The report enables you to:
- Formulate significant competitor information, analysis, and insights to improve R&D strategies.
- Identify emerging players with potentially strong product portfolio and create effective counter strategies to gain a competitive advantage.
- Identify and understand important and diverse types of pedicle screw-based dynamic stabilization systems under development.
- Develop market-entry and market-expansion strategies.
- Plan mergers and acquisitions effectively by identifying major players with the most promising pipeline.
- In-depth analysis of the product’s current stage of development, territory, and estimated launch date.
Colorado State University
Curtin University
Empirical Spine Inc
Jemo Spine, LLC.
Medtronic Plc
MI4 Spine, LLC
MicroPort Scientific Corp
NeuroPro Technologies Inc
NEXUS TDR Inc
NGMedical GmbH
Orthonex LLC (Inactive)
Paramount Surgicals Inc
Polytechnique Montreal
S14 Implants
SpineLab AG
X-spine Systems Inc
Table of Contents
Table
Figures
Frequently asked questions
-
Which territory has the highest number of products in the pedicle screw-based dynamic stabilization systems pipeline market?
As of November 2023, the US has the highest number of products in the pedicle screw-based dynamic stabilization systems pipeline market.
-
Which is the most followed regulatory pathway in the pedicle screw-based dynamic stabilization systems pipeline market?
As of November 2023, 510(k) is the most followed pathway for pipeline products in the pedicle screw-based dynamic stabilization systems market.
-
Which are the major companies operating in the pedicle screw-based dynamic stabilization systems pipeline market?
Some of the key companies in the pedicle screw-based dynamic stabilization systems pipeline market are ARO Medical, LLC, Colorado State University, Curtin University, Jemo Spine, LLC, and Medtronic Plc among others.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.