Peripheral Embolic Protection Devices Pipeline by Stages of Development, Segments, Region, Regulatory Path and Key Companies
Powered by ![]()
All the vital news, analysis, and commentary curated by our industry experts.
Peripheral Embolic Protection Devices Pipeline Market Report Overview
Embolic Protection Device(s) EPDs are used to capture and remove plaque debris that are dislodged during interventional procedures. Peripheral Embolic Protection Devices (EPDs) used in the lower extremity, carotid and renal arteries have been accounted for. One unit refers to one EPD. Carotid Embolic Protection Devices and Lower Extremity Embolic Protection Devices are tracked under this segment. Angioplasty: Angioplasty is a minimally invasive interventional procedure that is used to surgically unblock an artery with excessive plaque build-up to restore normal blood flow. Embolic debris may become dislodged during angioplasty procedures.
The peripheral embolic protection devices pipeline market research report provides comprehensive information about the peripheral embolic protection devices pipeline products with a comparative analysis of the products at various stages of development and information about the clinical trials which are in progress.
| Key Segments | Carotid Embolic Protection Devices and Lower Extremity Embolic Protection Devices |
| Key Territories | The US, Europe, China, Australia, Canada, Hong Kong, and Israel |
| Key Regulatory Paths | 510(k), CE, NMPA, PMA, TGA, MDCO, and MDL |
| Leading Companies | Abbott Vascular Inc, AorticLab Sarl, APT Medical Inc, Artio Medical, Avantec Vascular Corp, Cardiovascular Systems Inc, Emboline, Inc., Filterlex Medical Ltd, Innovia LLC, Instylla Inc, Keystone Heart Ltd, Neurosonix Ltd., and Protembis GmbH |
| Enquire & Decide | Discover the perfect solution for your business needs. Enquire now and let us help you make an informed decision before making a purchase. |
Peripheral Embolic Protection Devices Pipeline Market Segments
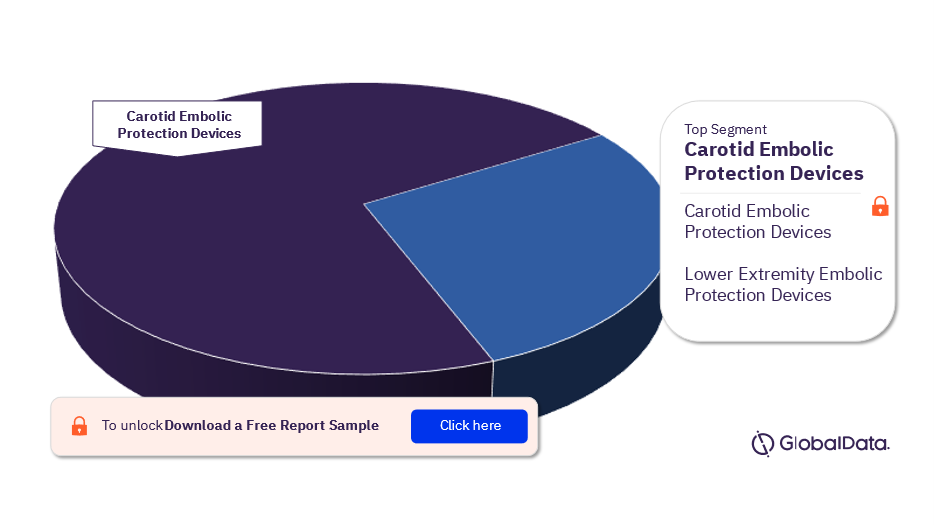
The key segments in the peripheral embolic protection devices market are Carotid Embolic Protection Devices and Lower Extremity Embolic Protection Devices. In 2023, the Carotid Embolic Protection Devices segment had the highest number of pipeline products.
Peripheral Embolic Protection Devices Pipeline Market Analysis by Segments, 2023 (%)
For more peripheral embolic protection devices market segments, download a free sample report
Peripheral Embolic Protection Devices Pipeline Market Segmentation by Territories
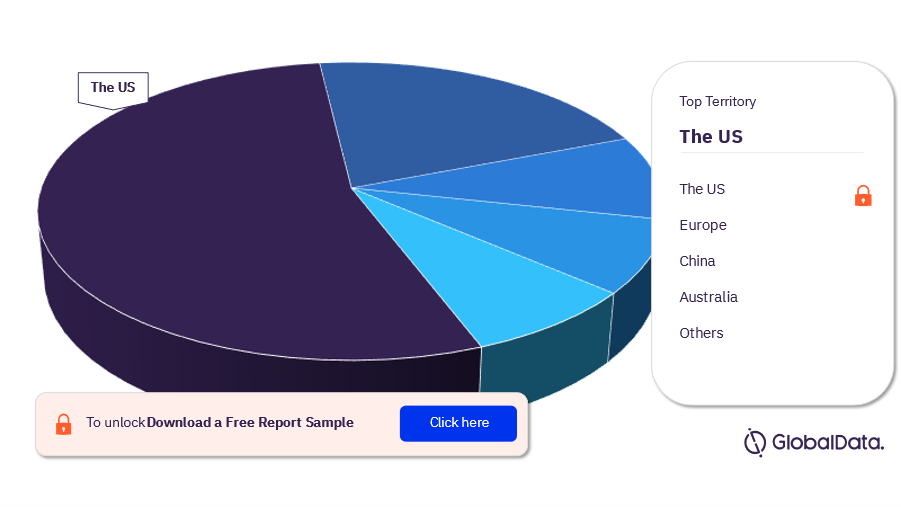
Some of the territories in the peripheral embolic protection devices market are the US, Europe, China, Australia, Canada, Hong Kong, and Israel among others. As of June 2023, the United States had the highest number of pipeline products.
Peripheral Embolic Protection Devices Pipeline Market Analysis, by Territories, 2023 (%)
For more territory insights into the peripheral embolic protection devices pipeline market, download a free report sample
Peripheral Embolic Protection Devices Pipeline Market Segmentation by Regulatory Paths
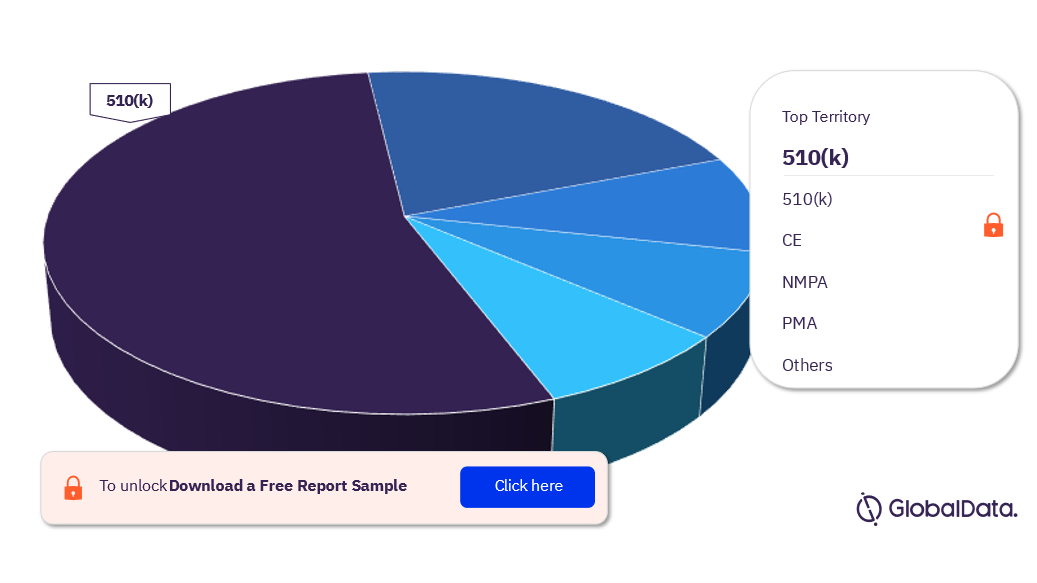
Some of the regulatory paths in the peripheral embolic protection devices pipeline market are 510(k), CE, NMPA, PMA, TGA, MDCO, and MDL among others. As of June 2023, 510(k) was the most followed pathway for pipeline products.
Peripheral Embolic Protection Devices Pipeline Market Analysis, by Regulatory Paths, 2023 (%)
For more regulatory path insights into the peripheral embolic protection devices pipeline market, download a free report sample
Peripheral Embolic Protection Devices Pipeline Market – Competitive Landscape
Some of the key companies in the peripheral embolic protection devices pipeline market are Abbott Vascular Inc, AorticLab Sarl, APT Medical Inc, Artio Medical, Avantec Vascular Corp, Cardiovascular Systems Inc, Emboline, Inc., Filterlex Medical Ltd, Innovia LLC, Instylla Inc, Keystone Heart Ltd, Neurosonix Ltd., and Protembis GmbH among others.
Abbott Vascular Inc: The Abbott Vascular Inc (Abbott Vascular), formerly Perclose, Inc., a subsidiary of Abbott Laboratories, is a medical device company that develops manufactures and markets cardiovascular devices. The company offers coronary intervention, peripheral intervention, and structural heart products. Its coronary intervention products include bleedback control valve, coronary dilatation catheters, coronary drug eluting stents, coronary guide wires, and others. Abbott Vascular’s peripheral intervention products comprise peripheral guide wires, dilatation catheters, guiding catheters, and stent grafts. The company provides minimally invasive treatment for mitral regurgitation and other structural defects of the heart. It also offers education and training services for healthcare professionals. Abbott Vascular is headquartered in Santa Clara, California, the US.
Segments Covered in the Report
Peripheral Embolic Protection Devices Pipeline Market Segment Outlook
- Carotid Embolic Protection Devices
- Lower Extremity Embolic Protection Devices
Peripheral Embolic Protection Devices Pipeline Market Territories Outlook
- The US
- Europe
- China
- Australia
- Canada
- Hong Kong
- Israel
Peripheral Embolic Protection Devices Pipeline Market Regulatory Paths Outlook
- 510(k)
- CE
- NMPA
- PMA
- TGA
- MDCO
- MDL
Scope
- Extensive coverage of the peripheral embolic protection devices under development
- Reviews details of major pipeline products which include, product description, licensing and collaboration details, and other developmental activities
- Reviews the major players involved in the development of peripheral embolic protection devices and list all their pipeline projects
- The coverage of pipeline products based on various stages of development ranging from Early Development to Approved / Issued stage
- The report provides key clinical trial data of ongoing trials specific to pipeline products
- Recent developments in the segment/industry
Reasons to Buy
The report enables you to:
- Formulate significant competitor information, analysis, and insights to improve R&D strategies
- Identify emerging players with the potentially strong product portfolios and create effective counter strategies to gain a competitive advantage
- Identify and understand important and diverse types of peripheral embolic protection devices under development
- Develop market-entry and market-expansion strategies
- Plan mergers and acquisitions effectively by identifying major players with the most promising pipeline
- In-depth analysis of the product’s current stage of development, territory, and estimated launch date
AorticLab Sarl
APT Medical Inc
Artio Medical
Avantec Vascular Corp
Cardio-Flow Ltd.
Cardiovascular Systems Inc
Emboline, Inc.
Ev3 Inc
Filterlex Medical Ltd
HeMo Bioengineering Ltd
Innovative Cardiovascular Solutions, LLC
Innovia LLC
Instylla Inc
Javelin Medical Ltd
Keystone Heart Ltd
Marblehead Medical LLC
MicroPort CardioFlow Medtech Corp
MicroPort Scientific Corp
MIV Therapeutics Inc
Neurosonix Ltd.
NewMed Medical Co Ltd
Protembis GmbH
University of Minnesota
Table of Contents
Table
Figures
Frequently asked questions
-
Which was the leading segment of the peripheral embolic protection devices pipeline market?
The Carotid Embolic Protection Devices segment had the highest number of pipeline products in 2023.
-
Which territory has the highest number of pipeline products in the peripheral embolic protection devices pipeline market as of June 2023?
As of June 2023, the US had the highest number of pipeline products in the peripheral embolic protection devices pipeline market.
-
Which is the most followed regulatory pathway in the peripheral embolic protection devices pipeline market as of June 2023?
As of June 2023, 510(k) was the most followed regulatory pathway in the peripheral embolic protection devices pipeline market.
-
Who are the major players operating in the peripheral embolic protection devices pipeline market?
Some of the major players operating in the peripheral embolic protection devices pipeline market are Abbott Vascular Inc, AorticLab Sarl, APT Medical Inc, Artio Medical, Avantec Vascular Corp, Cardiovascular Systems Inc, Emboline, Inc., Filterlex Medical Ltd, Innovia LLC, Instylla Inc, Keystone Heart Ltd, Neurosonix Ltd., and Protembis GmbH.
Get in touch to find out about multi-purchase discounts
reportstore@globaldata.com
Tel +44 20 7947 2745
Every customer’s requirement is unique. With over 220,000 construction projects tracked, we can create a tailored dataset for you based on the types of projects you are looking for. Please get in touch with your specific requirements and we can send you a quote.
Related reports
View more Peripheral Embolic Protection Devices reports