
Medical Devices DECODED
Previous edition: 13 May 2024
Share article
Get the full version straight to your inbox.
Exclusive access to our best-in-class data & intelligence
Subscribe now
Field Medical begins ventricular PFA technology trial
The FieldForce Ablation System is intended for the treatment of life-threatening ventricular arrhythmias.
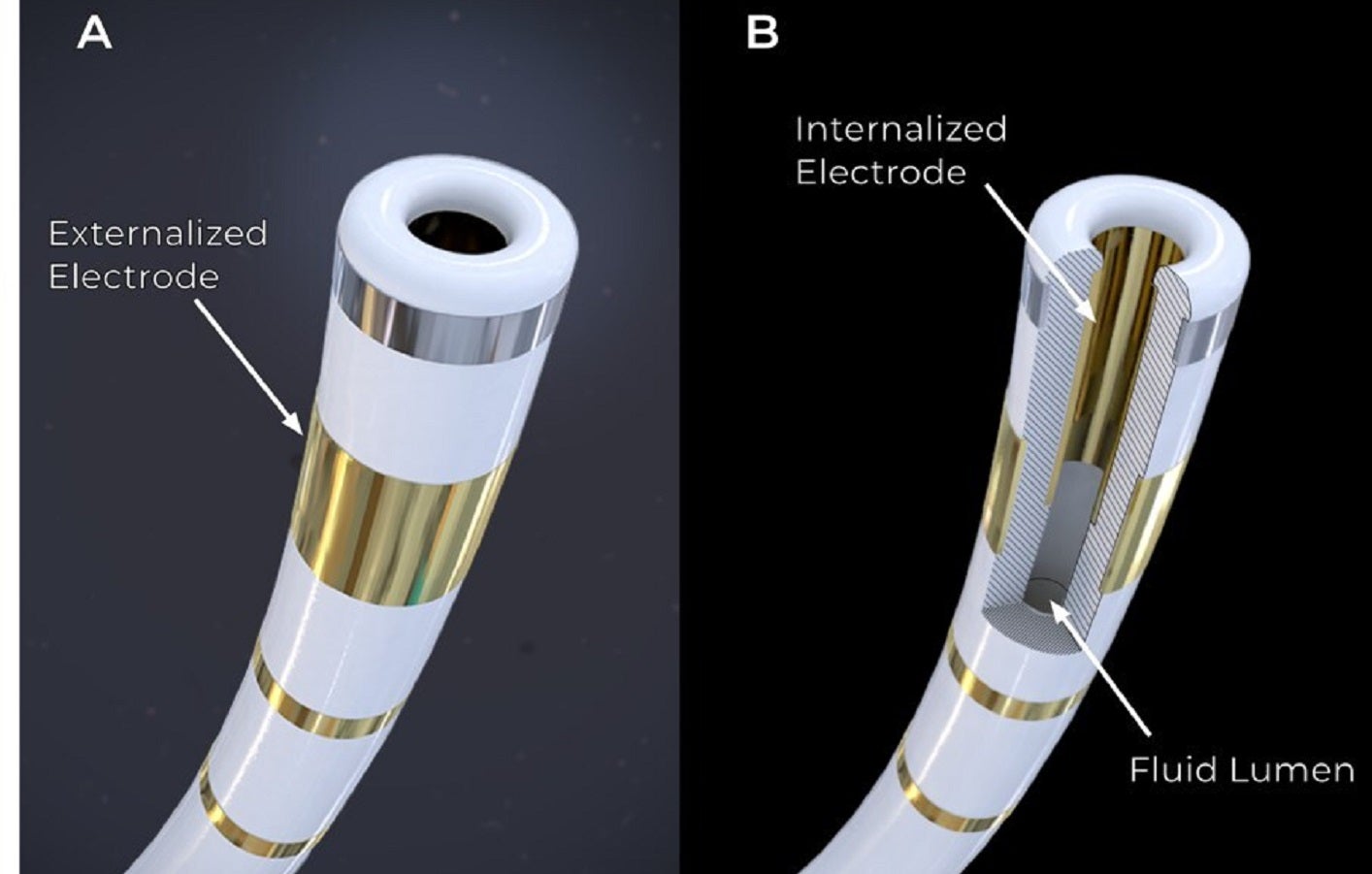
Field Medical has announced the commencement of first-in-human trials for its FieldForce pulsed field ablation (PFA) system at Na Homolce Hospital in Prague, Czech Republic.
FieldForce, which is said to be the first and only contact force PFA system, is designed for the treatment of life-threatening ventricular arrhythmias such as ventricular tachycardia (VT) and premature ventricular contractions (PVCs).
As part of the Ventricular Catheter Ablation Study (VCAS), the company will enrol up to 60 patients across five global sites.
This study is pivotal in demonstrating the safety and performance of the FieldForce ablation system, which aims to overcome the limitations of current PFA and radiofrequency ablation systems.
Field Medical CEO Steven Mickelsen said: “Our vision at Field Medical is to transform VT ablation into a widely available, one-hour outpatient procedure with improved safety outcomes.
“With the promising early data for our VCAS study, in much the same way that PFA transformed AF, the FieldForce ablation system is optimised to transform how ventricular arrhythmias are treated, offering hope to millions of patients worldwide.”
Field Medical's PFA technology offers a new solution with the first contact force PFA catheter optimised for the ventricle, potentially providing a predictable, time-efficient, and safer treatment option.
The VCAS study is a prospective safety and feasibility trial assessing the FieldForce Ablation System in patients with ventricular arrhythmias.
It is divided into two groups: VCAS-I, focusing on VT patients, and VCAS-II, targeting those with frequent PVCs.
Last year, Field Medical closed an oversubscribed seed round totalling $14m to help advance its catheter ablation platform.
Unnamed private investors led the convertible note investment, which aimed to support the company’s clinical development of the FieldBending technology.
Latest news

Dementia Action Week 2024: the future of Alzheimer's diagnostics
As the debate over using tau levels to guide patient treatment persists, the landscape for Alzheimer’s diagnostics continues to move ahead.

Siemens Healthineers announces £250m UK MRI facility
The Siemens Healthineers facility is designed to address a critical shortage in helium and is aimed at providing 1,300 jobs in the industry.
FDA approval paves way for Masimo's debut in OTC baby monitoring sector
The shift towards remote healthcare and increased interest in health monitoring at home has driven demand for advanced healthcare technologies, including baby monitoring systems.

Triomics wins $15m for oncology GenAI software
Triomics's GenAI platform helps cancer centres streamline administrative tasks.

Industry takes: the importance of keeping healthcare businesses cybersecure
Cybersecurity is vital for all industries, but there are in which breaches have more impact than healthcare.

UK NICE backs Exact's Oncotype cancer genomic test
The UK agency has recommended expanded use of Exact’s Oncotype DX test to help physicians make chemotherapy decisions for breast cancer patients.
In our previous edition

Medical Devices Decoded
Neuralink's first brain chip implant faces setback
10 May 2024

Medical Devices Decoded
FDA issues warning on Getinge device amid quality concerns
09 May 2024

Medical Devices Decoded
Samsung to acquire foetal ultrasound AI software company Sonio
08 May 2024
Newsletters in other sectors
Aerospace, Defence & Security
Automotive
Consumer
Medical Devices
Travel and Tourism
Search companies, themes, reports, as well as actionable data & insights spanning 22 global industries
Access more premium companies when you subscribe to Explorer


