
Medical Devices DECODED
Previous edition: 13 May 2024
Share article
Get the full version straight to your inbox.
Exclusive access to our best-in-class data & intelligence
Subscribe now
Siemens Healthineers announces £250m UK MRI facility
The Siemens Healthineers facility is designed to address a critical shortage in helium and is aimed at providing 1,300 jobs in the industry.

Medical device giant Siemens Healthineers has announced a $250m investment in a new UK production facility aimed at manufacturing superconducting magnets to be used in Magnetic Resonance Imaging (MRI) devices.
Siemens Healthineers has said that the North Oxfordshire facility will both support more than 1,300 jobs in the area and minimise the use of limited and unsustainable materials in making MRIs, such as helium. The facility will be the UK’s first-ever home for what the company calls DryCool technology, which reduces the amount of helium required in an MRI scanner from 1,500 litres to under a single litre.
At the same time, the German medical device firm has said that the 56,000m2 site will be operationally carbon-neutral with plans to open its doors in 2026. The new site will help bolster the MRI supply amid a rising global demand for MRI products as populations age and chronic diseases become more prevalent.
Chief executive of Siemens Healthineers, Bernd Montag said: “MRI technology plays a vital role in diagnosing disease, helping patients to get healthy and stay healthy. As a world leader in medical imaging, we are very proud to open the next chapter of our history here in Oxford. This factory will be the global centre for our innovative low-helium magnet technology, meaning we consume far less of a scarce natural resource and enable access to MRIs for many more patients.”
Additionally, the new facility will act as a research and development hub responsible for technologies such as designing and manufacturing some of what Siemens calls the world’s smallest and most lightweight whole-body scanners.
Commenting on the opening of the new facility, UK Prime Minister Rishi Sunak, said: “I am delighted Siemens Healthineers has chosen Oxfordshire for its new facility, supporting over 1,300 skilled jobs in the area, and reinforcing the region’s status as a vanguard in healthcare and R&D. But as well as the incredible local benefits this will bring, this cutting-edge facility also presents an opportunity to enhance patient care globally – which means British innovation is saving lives around the world.”
It follows after German-multinational company announced plans to close its fast-track diagnostics arm, putting 90 jobs at risk and pivoting away from a Covid-19 era business. It also follows after the company received approval from the US Food and Drug Administration (FDA) for its MAGNETOM Cima.X 3 Tesla MRI device.
Latest news

Dementia Action Week 2024: the future of Alzheimer's diagnostics
As the debate over using tau levels to guide patient treatment persists, the landscape for Alzheimer’s diagnostics continues to move ahead.
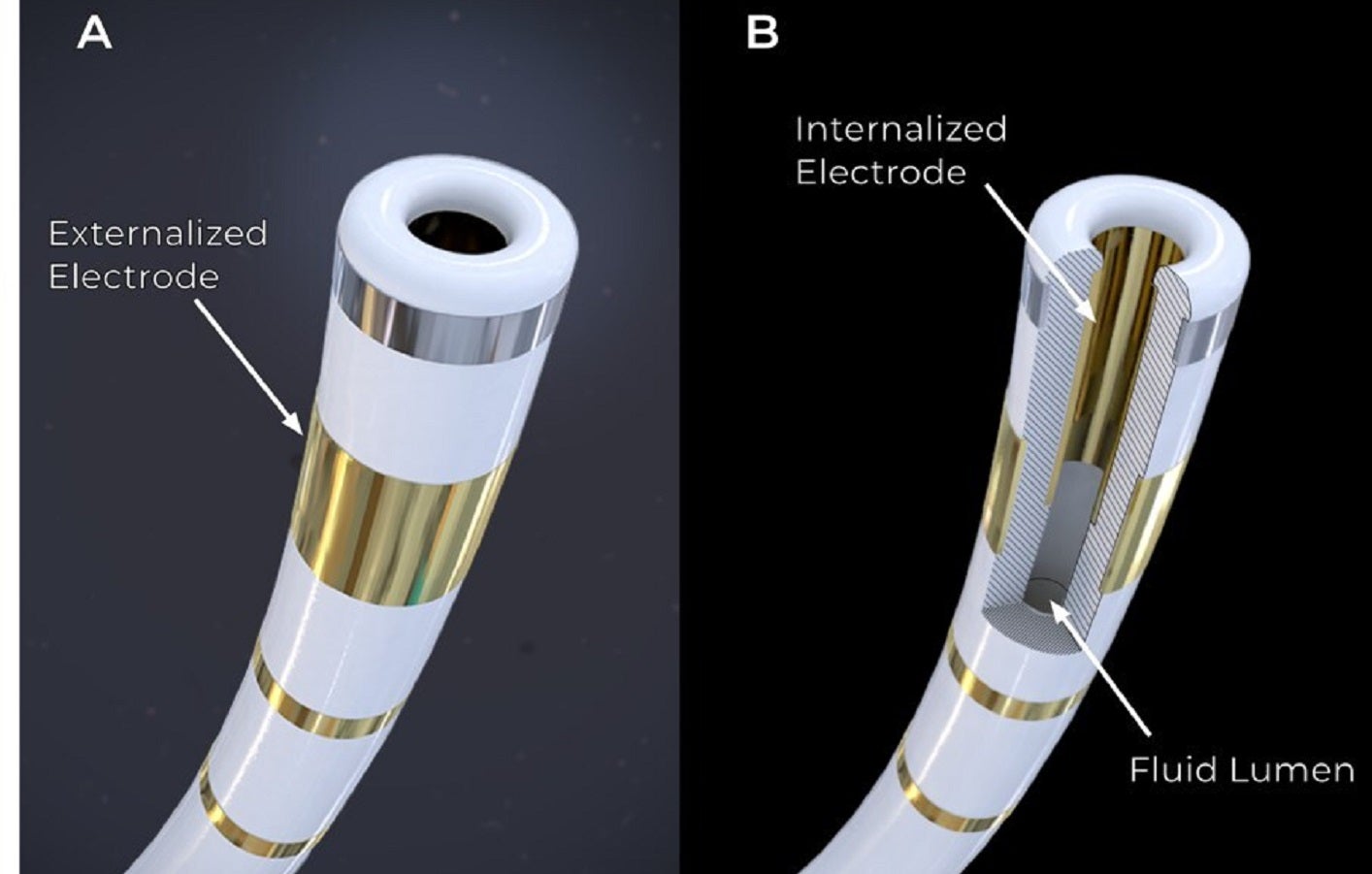
Field Medical begins ventricular PFA technology trial
The FieldForce Ablation System is intended for the treatment of life-threatening ventricular arrhythmias.
FDA approval paves way for Masimo's debut in OTC baby monitoring sector
The shift towards remote healthcare and increased interest in health monitoring at home has driven demand for advanced healthcare technologies, including baby monitoring systems.

Triomics wins $15m for oncology GenAI software
Triomics's GenAI platform helps cancer centres streamline administrative tasks.

Industry takes: the importance of keeping healthcare businesses cybersecure
Cybersecurity is vital for all industries, but there are in which breaches have more impact than healthcare.

UK NICE backs Exact's Oncotype cancer genomic test
The UK agency has recommended expanded use of Exact’s Oncotype DX test to help physicians make chemotherapy decisions for breast cancer patients.
In our previous edition

Medical Devices Decoded
Neuralink's first brain chip implant faces setback
10 May 2024

Medical Devices Decoded
FDA issues warning on Getinge device amid quality concerns
09 May 2024

Medical Devices Decoded
Samsung to acquire foetal ultrasound AI software company Sonio
08 May 2024
Newsletters in other sectors
Aerospace, Defence & Security
Automotive
Consumer
Medical Devices
Travel and Tourism
Search companies, themes, reports, as well as actionable data & insights spanning 22 global industries
Access more premium companies when you subscribe to Explorer


